38 nacl dot diagram
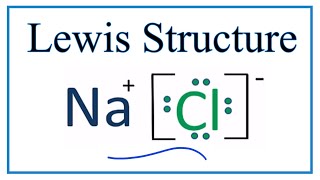
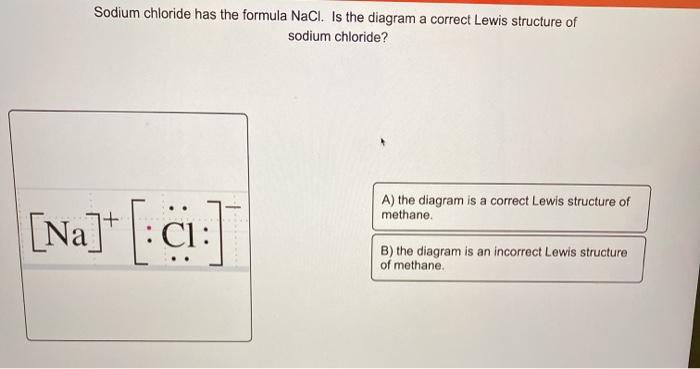
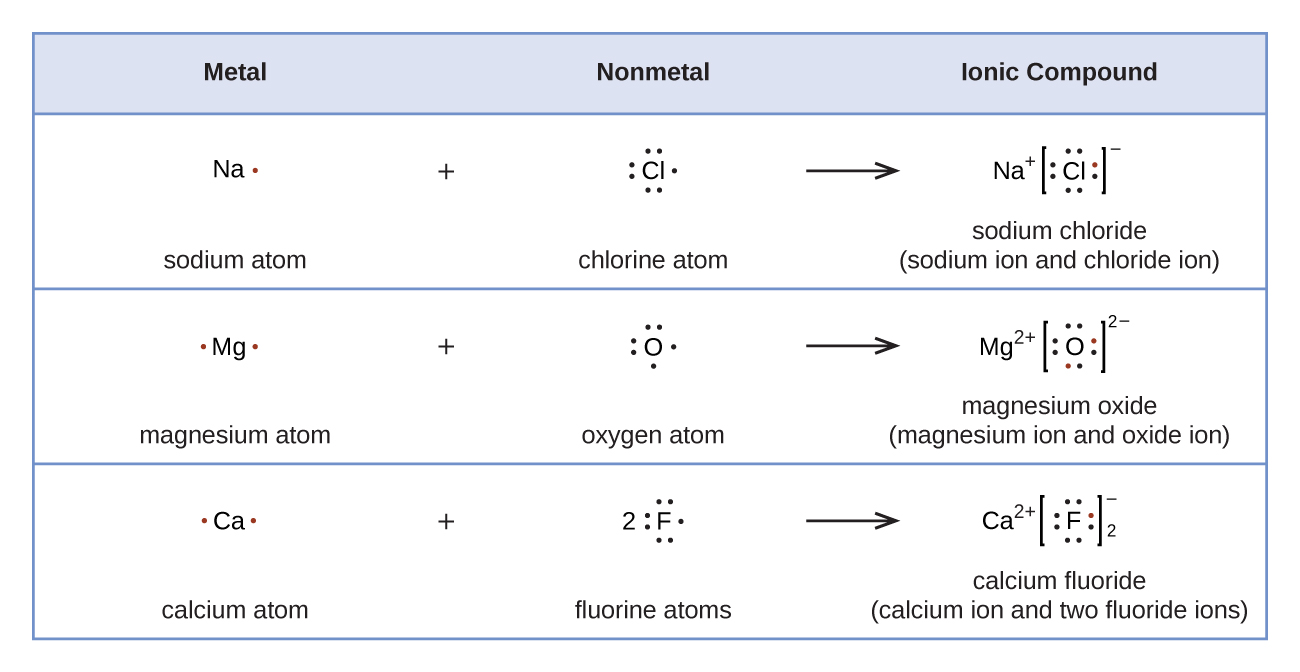
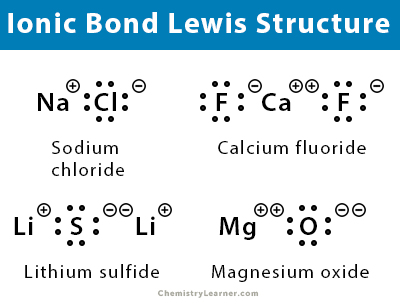
For example, consider sodium chloride. The Lewis Structure for the Salt NaCl, shows two ions which have their (Now) outer shells of electrons filled with a complete octet. In the case of the sodium cation, the filled shell is the outermost of the 'core' electron shells. ... Lewis dot diagrams give us a static picture of what the molecule or ion ... The Lewis dot structure of NaCl consists of a chloride ion surrounded by eight electron dots (four pairs) and a sodium ion bonded to that chlorine ion. Typically, ionic Lewis dot structures include the ionic charge, so the Na ion is labeled +1 and Cl is labeled -1.
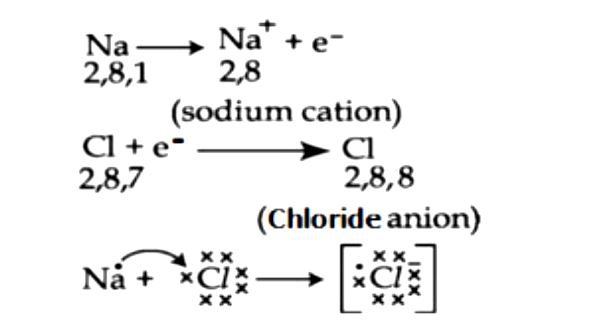
Diagram of bonding in sodium chloride. A sodium atom gives an electron to a chlorine atom. The result is a sodium ion (2,8)+ and a chloride. In Ionic Bonds valence electrons are completely transferred (not shared). Thus, we write the Lewis structure for NaCl as: NaClLewisDot.
Nacl dot diagram
For example to draw a dot and cross diagram of sodium chloride NaCl we draw a sodium ion Na next to a chloride ion Cl -. In a dot and cross diagram. For example the dot and cross diagram below shows the molecule chlorine monofluoride which is made up of a chlorine atom and a fluorine atom joined by a single covalent bond. The following electron dot diagram shows the formation of NaCl. Formation of MgF 2 The following electron dot diagram shows the formation of MgF 2. Notice that instead of drawing electron dot diagrams for 2 F-, the number “2” has been placed in front of one of the F-. 3 Check me out: http://www.chemistnate.com
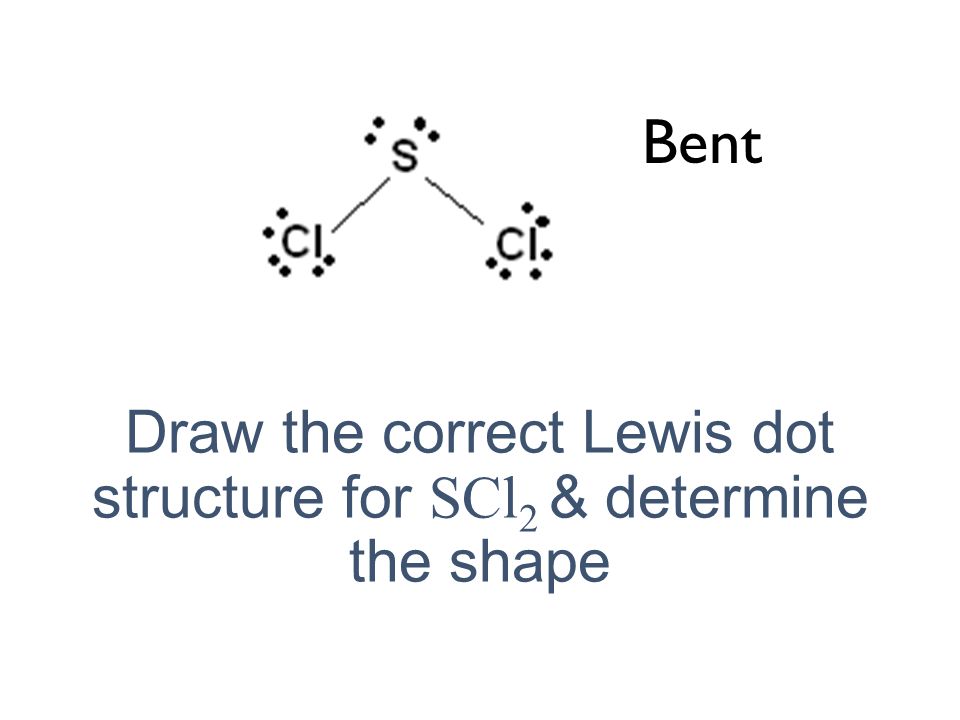
Nacl dot diagram. Sodium, a metal in Group 1, loses one electron to become a +1 ionChlorine, a non-metal in Group 17, gains one electron to become a -1 ion.Together, they comb... Dot and cross diagrams help us to model when ions are formed from atoms. Here's an example using sodium and chlorine. They form ions which bond to form sodium chloride. 1. Draw the electronic ... one of the oxygen atoms. In the box below, draw a Lewis electron-dot diagram (or diagrams) for the. HCO 3 − ion that is (are) consistent with the given information. (f) A student prepares a solution containing equimolar amounts of. HC 2 H 3 O 2 and NaC 2 H 3 O 2. The . pH of . the solution is measured to be . 4.7. The student adds two drops ... To draw a dot and cross diagram of an ionic compound, we simply draw dot and cross diagrams of the ions involved, in the correct ratio. For example, to draw a dot and cross diagram of sodium chloride (NaCl), we draw a sodium ion (Na +) next to a chloride ion (Cl-). Because the ratio of sodium to chloride is 1:1, we draw one of each ion:
It is represented by dots in the NaCl Lewis diagram. The NaCl molecule's core chlorine atom can be represented as follows: Total outermost valence shell electron of chlorine atom in NaCl= 7 Total outermost valence shell electron of sodium atom in NaCl= 1 The NaCl molecule has one central chlorine and one sodium atoms. Draw Orbit Structure and Electron Dot Diagram of Naci, Mgcl2 and Cao. CBSE CBSE (English Medium) Class 10. Question Papers 886. Textbook Solutions 18445. MCQ ... Orbit structure and electron dot diagram of NaCl: Orbit structure and electron dot diagram of MgCl2: Orbit structure and electron dot diagram of CaO: Concept: Electrovalent (or Ionic ... Sodium Chloride Dot And Cross - 9 images - igcse chemistry, dot cross diagram aluminium oxide youtube, What is the electron dot structure for NaCl? Sodium atom will loose one electron to gain noble gas configuration and form sodium cation with +1 charge. Chlorine atom will gain one electron to gain noble gas configuration and form chloride ion with -1 charge. In sodium chloride the one electron from sodium metal gets transferred to chlorine atom.
A step-by-step explanation of how to draw the NaCl Lewis Dot Structure.For the NaCl Lewis structure, calculate the total number of valence electrons for the ... Draw electron dot representation for the formation of sodium chloride. Solve Study Textbooks. >>. Class 11. >> Chemistry. >> Chemical Bonding and Molecular Structure. The Lewis dot structure of NaCl consists of a chloride ion surrounded by eight electron dots (four pairs) and a sodium ion bonded to that chlorine ion. Thus, to determine the oxidation number of the two atoms, we count the electrons It is important to keep the formal charges as low as possible. and hence several thousands times slower than electrons). Complete the dot and cross diagram on Figure 4 ... (NaCl). Figure 6 Explain the conditions needed for sodium chloride to conduct electricity. [3 marks]
Above is the electronic dot & cross diagram for the ionic bonding in the ionic compound sodium chloride, is the Lewis diagram for the formation of sodium chloride from its elements. simplified Limitations of these dot and cross diagrams:
A step-by-step explanation of how to draw the NaCl Lewis Dot Structure (Sodium chloride).For NaCl we have an ionic compound and we need to take that into acc...
Sodium Chloride Dot Diagram. Here are a number of highest rated Sodium Chloride Dot Diagram pictures on internet. We identified it from reliable source. Its submitted by paperwork in the best field. We assume this nice of Sodium Chloride Dot Diagram graphic could possibly be the most trending subject later we allowance it in google benefit or ...
The lewis dot structure of NaCl contains one positive charge on sodium metal and one negative charge on chlorine nonmetal. We have to represent them by putting brackets around them. The lewis diagram of an ionic compound is formed with a different approach than the normal procedure we used for drawing the compounds like NH3, BF3, BrF5, etc.
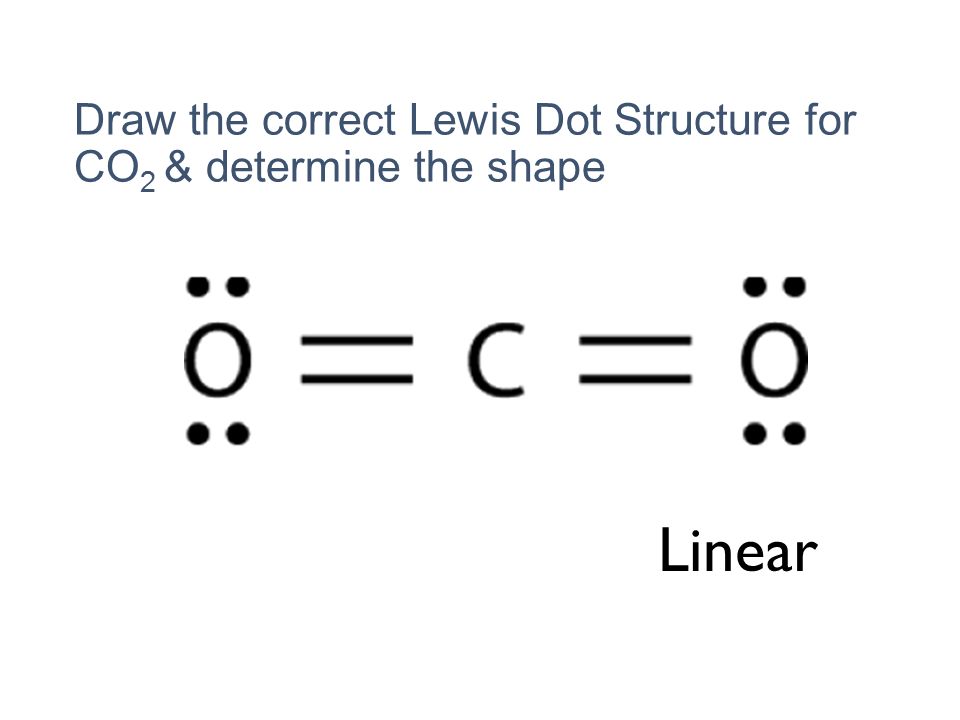
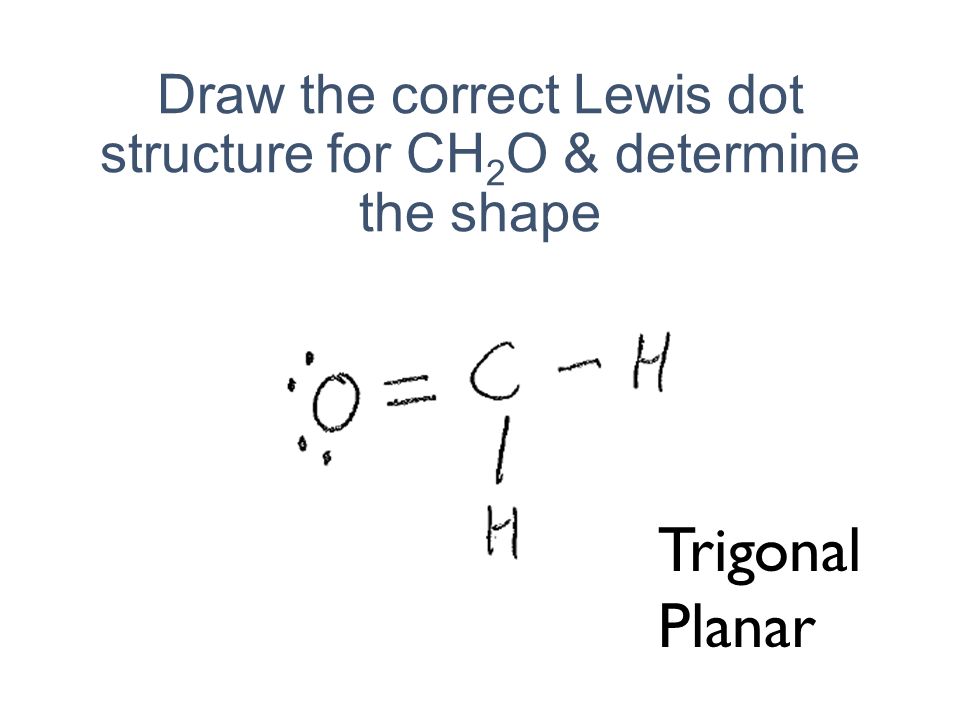
Lewis Structure Examples. The Lewis electron dot structures of a few molecules are illustrated in this subsection. 1. Lewis Structure of CO2. The central atom of this molecule is carbon. Oxygen contains 6 valence electrons which form 2 lone pairs. Since it is bonded to only one carbon atom, it must form a double bond.
Lewis electron dot symbol to describe ionic bonding in sodium chloride. The formation of sodium chloride involves sodium metal, Na and chlorine gas, Cl 2. The reaction can be thought of involving the following simultaneous processes: The oxidation of sodium metal. Sodium atoms lose its outer-shell electron to become a sodium ion, (i.e. cation).
Sodium Chloride Lewis Dot Structure December 6, 2021 by admin 1 View Ionic Bonding In Sodium Chloride When Only The Outer Electron Shells Are Drawn Ionic Bonding Tech Company Logos Vimeo Logo
Sodium Chloride is the chemical name of NaCl. The NaCl Molecular Weight (Sodium Chloride) is 58.44 g/mol. Visit BYJU'S to understand the properties, structure, and uses of Sodium Chloride (NaCl) explained by India's best teachers.
The dot diagram only shows electrons in the atom's outermost energy level. No electrons are shown since the only electron in the outermost energy level of sodium was transferred to chlorine. In the final dot diagram of NaCl, the dots between the sodium and chlorine are between the atoms.
Dot and cross diagrams. ... Ionic bonding in sodium chloride Question. Draw a diagram, with outer electrons only, to show how the electrons are transferred when magnesium chloride is formed from ...
Check me out: http://www.chemistnate.com
The following electron dot diagram shows the formation of NaCl. Formation of MgF 2 The following electron dot diagram shows the formation of MgF 2. Notice that instead of drawing electron dot diagrams for 2 F-, the number “2” has been placed in front of one of the F-. 3
For example to draw a dot and cross diagram of sodium chloride NaCl we draw a sodium ion Na next to a chloride ion Cl -. In a dot and cross diagram. For example the dot and cross diagram below shows the molecule chlorine monofluoride which is made up of a chlorine atom and a fluorine atom joined by a single covalent bond.












.jpg)









Comments
Post a Comment