38 energy level diagram for hydrogen atom
Draw an atom of Hydrogen, to the proper scale., Draw a diagram that accurately reflects how the Plum Pudding model describes an atom, What experiment led to the concept of the nuclear atom?, Who is the first person to propose the idea that matter is made up of small, indivisible particles, according to historical records? These particles are electrically charged, and the electric forces on the charge are responsible for holding the atom together. Attempts to separate these smaller... They can be created only with the addition of enormous amounts of energy, however, and are very short-lived. All atoms are roughly the same size, whether they have...
Hydrogen atom has no outer energy level of electrons making the single proton in the nucleus unusually bare. The proton is thus readily available for any form of dipole-dipole attractions. The bare proton of the hydrogen atom thus attracts the more electronegative atom (e.g. N, O and F) on either side.
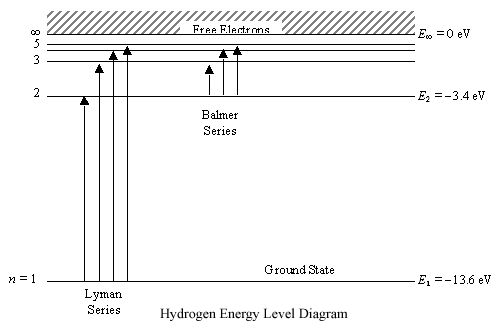
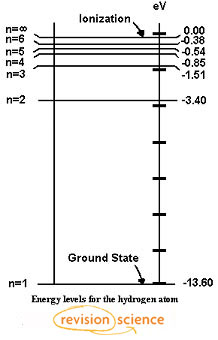
Energy level diagram for hydrogen atom
The grand challenge in the development of atomically dispersed metallic catalysts is their low metal-atom loading density, uncontrollable localization and... catalytic behaviour is associated with an ensemble effect via the synergy of Co adatom and S of the D-1T MoS 2 support by tuning hydrogen binding mode at the interface. Copper Atom Diagram. Energy Level. Hydrogen Atomic Orbital. Insulator Electrons. Electron Hole. Free Electron Model. Silicon Valence Electrons. Electron Sea Model. Manganese (Mn) excited state electron configuration and orbital diagram. When a manganese atom is excited, then the manganese atom absorbs energy. As a result, an electron in the 4s orbital jumps to the 4p x sub-orbital. The p-orbital has three sub-orbitals. The sub-orbitals are p x, p y, and p z.
Energy level diagram for hydrogen atom. Normally I say that the F atom has a higher electronegativity than the H atom, which causes a net positive dipole moment. However now I am trying to use the MO diagram. My rough guess is that because the H atom has one of its electrons moving down to the energy level, it is effectively showing that it is less electronegative? Use the molecular orbital energy level diagram to show that N 2 would be expected to have a triple bond, F 2, a single bond and Ne 2, no bond. Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. “But to really see what was happening at the single-atom level we had to use a uniquely sensitive atomic force microscope in Michael Crommie’s laboratory.”... The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit. DOE’s Office of Science is the single... to hydrogen gas (H 2) and carbon dioxide (CO 2), it could also lower the cost of running this so-called “water gas shift” reaction. “With low temperature and pressure, the energy consumption will be lower and the experimental setup will be less expensive and easier to use in small settings, like fuel cells for cars...
This is strictly true for the hydrogen atom where the energy levels depend only upon the principal quantum number fine structure neglected. Argon The Next 18. But in larger atoms the energy depends also upon the orbital quantum number so the sub-levels are filled in the order s p d f etc. He proved that electrons surrounding the atom do not revolve arbitrarily. Rather, they revolve in well-defined orbitals at very specific energy levels, i.e., the orbitals are quantized. Through this proof, Bohr's model of the atom was able to explain certain phenomenon, such as the hydrogen spectrum. However, that's a whole other discussion. the atom itself (using light of short wavelength) will, if repeated a large number of times, result in for . That is, the electron will be detected close to the... of energy required to ionize the hydrogen atom. If light with a wavelength comparable to the diameter of the atom is employed in the experiment, then the electron... File electron shell 019 potassium atomic radius the atomic radius of a chemical element is a measure of the size of its atoms usually the mean or typical distance from the center of the nucleus to the the rutherford bohr model of the hydrogen atom z 1 or a hydrogen like ion z 1 in this model it is an essential feature that the photon energy or.
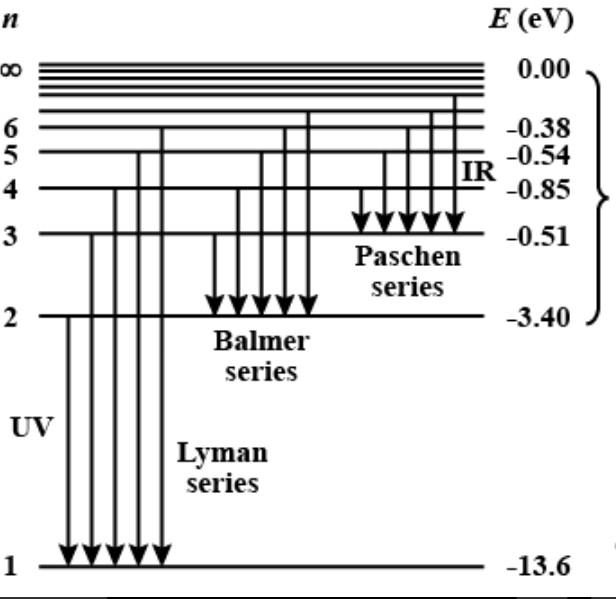
1. Introduction. Hydrogen energy as a new type of power is widely developed and applied at present, and the preparation of hydrogen is an important part of the hydrogen economy [, , , , ].Green, sustainable water electrolytic hydrogen generation is one of the most perspective methods of hydrogen production, and Platinum (Pt)-based catalysts are considered to be the most effective catalysts for ... 1. Orbital diagram of Hydrogen (H) 2. Orbital diagram of Helium (He) 3. Orbital diagram of Lithium (Li) 4. Orbital diagram of Beryllium (Be) 5. AUGUST 2018 OPPORTUNITIES FOR AUSTRALIA FROM HYDROGEN EXPORTS ACIL ALLEN CONSULTING FOR ARENA ACIL ALLEN CONSULTING PTY LTD ABN 68 102 652 148 LEVEL NINE 60... 1 PROJECTED DEMAND FOR HYDROGEN FOR ENERGY USE, BY CAGR A–5 FIGURE A.2 CHINA ENERGY CONSUMPTION 2014 A–9 FIGURE B.1 CALCULATION OF DIRECT VALUE ADDED B–2 FIGURE C.... In this section we will discuss the energy level of the electron of a hydrogen atom, and how it changes as the electron undergoes transition. According to Bohr's theory, electrons of an atom revolv...
Shell model is a nuclear model of the atomic nucleus. To explain the structure of the nuclear energy level, the shell model uses the principle of Pauli exclusion.Different nuclear shell models were proposed to study the complete nucleus of an atom structure. Dmitry Ivanenko in 1932 first introduced the shell model and later in 1949 this shell model was developed by different Physicists namely ...
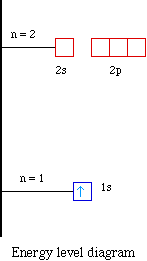
The hydrogen atom is unusual, however. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Within a given shell, the s orbitals always have the lowest energy. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.
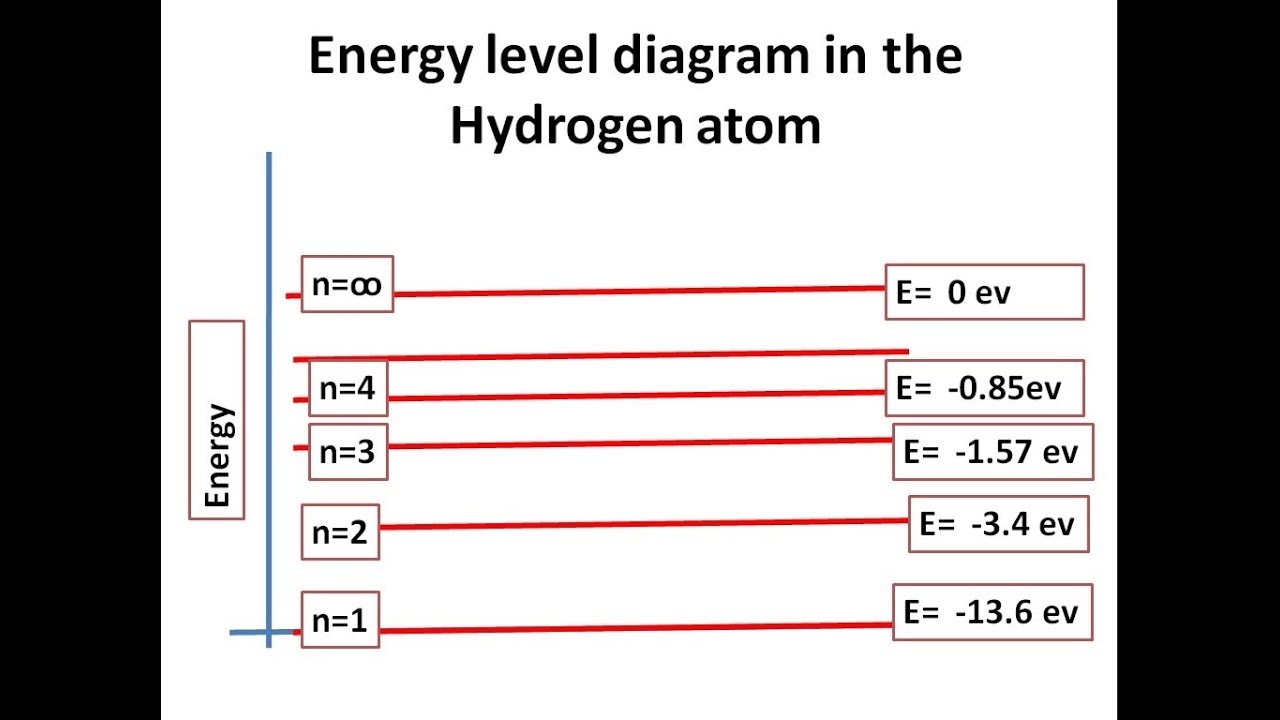
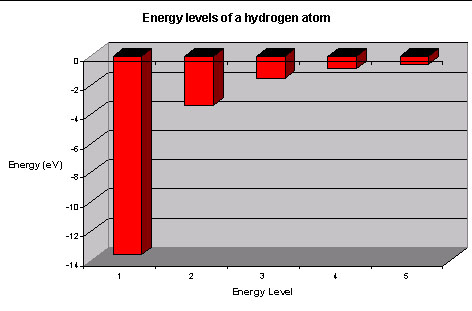
Sep 27, 2015 · A simple expression for the energy of an electron in the hydrogen atom is: #E=-(13.6)/(n^2)# where the energy is in electron volts #n# is the principle quantum number. This gives rise to the familiar electron energy level diagram where they converge and coalesce.
상세보기 목록 L23.1 Energy levels and diagram for hydrogen.외부학습자원 Type anywhere to search 전체 국정철학·과제 공직가치 분야별 UCC
An energy level can be measured by the amount of energy needed to unbind the electron from the atom, and is usually given in units of electronvolts (eV). The lowest energy state of a bound electron is called the ground state, i.e. stationary state, while an electron transition to a higher level results in an excited state.
An atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. 2. Protons and neutrons reside in the nucleus and are together called nucleons. 3. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. 4.
Primary energy is needed to create hydrogen fuel. This energy comes either from burning , using(which David S. Scott wrote about extensively), or by using sources like and . While it may not sound like anuse of energy to do this, hydrogen fuel is about ten times more by volume than Lithium-ion batteries, and more than 100x more...
What are the main conversion methods involved in using hydrogen for energy purposes? Alongside the mobility applica- tions for hydrogen technology, are there any stationary applications for hydrogen as a source of energy? The focus of this study is the issue of sustainable mobility through fuel cells and hydrogen (H 2 ). When...
The hydrogen atom has an energy level diagram as shown. What type of light is emitted from moving between each energy level? [Take Planck's Constant to be 4.136E-15 eVs.] (a) n=6 to n=2 (b) n=5 to n=2 (c) n=4 to n=3. Jan 13 2022 02:44 AM. Solution.pdf.
As a class, models of the hydrogen atom are explored using an online java applet in this activity. Real-time spectrometer outputs, visual representations, and energy level diagrams (where appropriate) of the following models are compared and contrasted: Billiard Ball, Plum Pudding, Classical Solar System, Bohr, deBroglie, and Schr ö dinger.
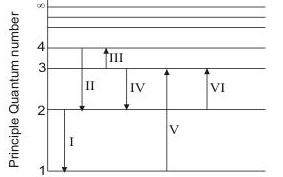
January 10, 2022 thanh. For This question consider the figure to the right shows an energy level diagram for a certain atom (not hydrogen). Several transitions are shown and are labeled by letters. Note: The diagram is not drawn to scale. Which transition corresponds to the absorption of the photon with the longest wavelength?
Match the type of system or situation to the appropriate energy level diagram. electronic states or a single atom such as hydrogen hadronic (such as delta +) nuclear (such as the nucleus of a carbon atom) electronic, vibrational, and rotational...
energy of an orbital formula. petal fair launch auction; energy of an orbital formula 26 January, 2022 ...
Ionization energy is positive for neutral atoms, meaning that the ionization is an endothermic process. Roughly speaking, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy. In physics, ionization energy is usually expressed in electronvolts (eV) or joules (J).
renewable energy source for the future. But in reality, the overwhelming majority of hydrogen fuel is obtained from... The NiCo-SAD-NC electrocatalyst had a comparable level of overvoltage as commercial Pt-based catalysts in acidic and alkaline media. NiCo-SAD-NC also exhibited eight times higher activity than Ni/Co single-atom...
May 21, 2021 · The p–d orbital hybridization between single Pt atom and coordinating atom (e.g., S and Se) on the support broadens the d level of single-atom Pt, leading to the formation of a narrower d band ...
Hydrogen Atom Energy Level Diagram. Gallery of Energy Level Wavelength. Dog Behaviorist Near Me Breakfast Catering Near Me Car Battery Near Me Seafood Places Near Me Organic Grocery Store Near Me Property Surveyor Near Me Cdl Classes Near Me Barro S Pizza Near Me Craigslist En Denver Co Cat Declawing Near Me Singing Lessons Near Me Lumber ...
It bonds to negative ions using hydrogen bonds.The diagram shows the potential hydrogen bonds formed with a chloride ion, Cl-. Although the lone pairs in the chloride ion are at the 3-level and would not normally be... the hydrogen atom has to be attached directly to the very electronegative element for hydrogen bonding to...
Bohr's model of the atom was based on the idea the angular momentum is quantized, and quantized in a particular way. de Broglie came up with an explanation for why the angular momentum might be quantized in this way. de Broglie realized that if you use the wavelength associated with the electron, and only allow for standing...
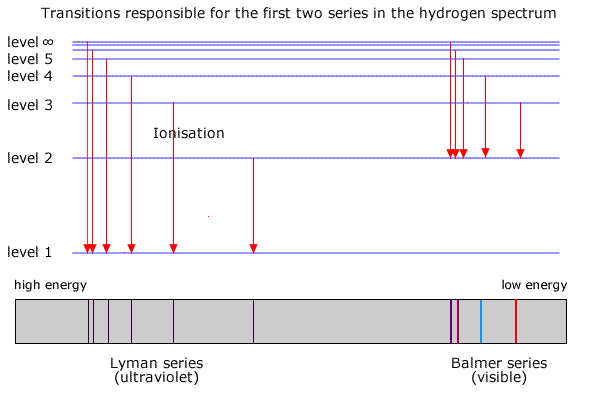
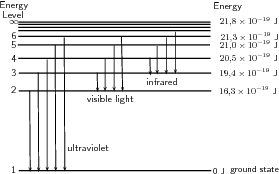
Mar 02, 2020 · Energy level diagrams indicate us the different series of lines observed in a spectrum of the hydrogen atom. The horizontal lines of the diagram indicate different energy levels. The vertical lines indicate the transition of an electron from a higher energy level to a lower energy level. It is very important that as indicated in the diagram ...
Asymmetry energy (also called Pauli Energy). An energy associated with the Pauli exclusion principle . Were it not for the Coulomb energy, the most stable form of nuclear matter would have the same number of neutrons as protons, since unequal numbers of neutrons and protons imply filling higher energy levels for one type of particle, while ...
The energy level of an atom's valence electrons correspond to its period or horizontal row on the periodic table. Hydrogen and helium, both in the first period, have their valence electrons in the. Derive the predicted ground-state electron configurations of atoms; Identify and explain exceptions to predicted electron configurations for atoms ...
nanomaterials for hydrogen evolution reaction (HER), yet the simplicity of single-atom center limits its further modification and utilization. Obtaining bimetallic single-atom-dimer (SAD) structures can reform the electronic structure of SACs with added atomic-level synergistic effect, further improving...
The Co sites on Pt 1.5 Co 0.08 and [email protected] 1.5 Co 0.08 surface exhibit stronger affinity to the electron-enriched O atom in H 2 O molecule than Pt surface, originating from the less filled d-orbitals of Co (3d 7 4s 2) compared to Pt (5d 9 6s 1), which results in enhanced adsorption energy of H 2 O and reduced energy barrier for water ...
Manganese (Mn) excited state electron configuration and orbital diagram. When a manganese atom is excited, then the manganese atom absorbs energy. As a result, an electron in the 4s orbital jumps to the 4p x sub-orbital. The p-orbital has three sub-orbitals. The sub-orbitals are p x, p y, and p z.
Copper Atom Diagram. Energy Level. Hydrogen Atomic Orbital. Insulator Electrons. Electron Hole. Free Electron Model. Silicon Valence Electrons. Electron Sea Model.
The grand challenge in the development of atomically dispersed metallic catalysts is their low metal-atom loading density, uncontrollable localization and... catalytic behaviour is associated with an ensemble effect via the synergy of Co adatom and S of the D-1T MoS 2 support by tuning hydrogen binding mode at the interface.





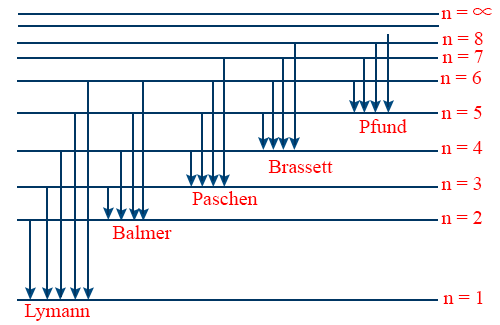
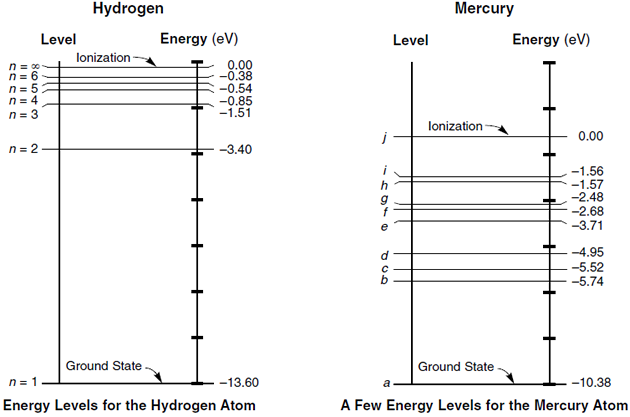


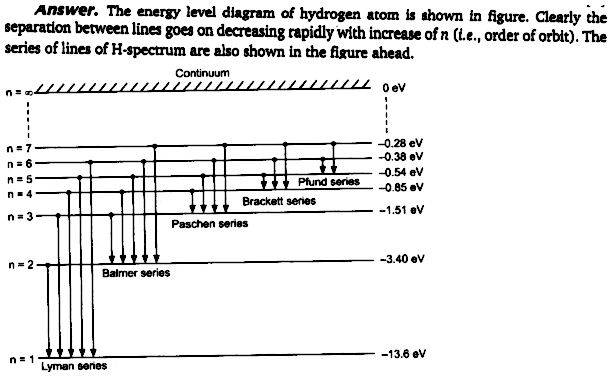












Comments
Post a Comment