39 orbital diagram nitrogen
The hybridization of the central Nitrogen atom is sp2. Molecular Orbital (MO) Diagram. In VBT, also known as Valence Bond Theory, we consider the fact that atomic orbitals ( AOs ) from the same individual atom can come together to form fusion into hybridized orbitals which overlap with hybridized orbitals formed by the combination of AOs from ... 37) The orbital diagram for fluorine shows 1 unpaired electron in a p orbital. 38) The correct electron configuration for magnesium is: 1s 2 2s 2 2p 6 3s 3 . 39) The element manganese (symbol = Mn) has five valence electrons.
Molecular orbital s: Orbital s that span two or more atoms. These are constructed by overlapping atomic orbital s (AOs) which match in symmetry and size. In principle, To construct MO diagram of a any Molecule, first, set up Schrödinger wave equation for that molecule and then, solve it!!! Molecular Orbital Diagram Maker. These quizzes enable you to build your own molecular orbital diagram ...

Orbital diagram nitrogen
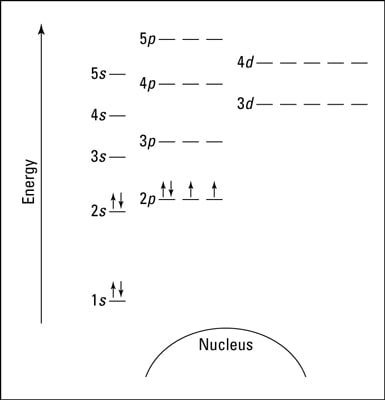
An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. A box, line, or circle, is drawn to represent each orbital in the electron configuration (using the Aufau Principle to order the orbitals and hence the boxes, lines or circles, as shown below) Molecular orbital diagram for hydrogen: For a diatomic molecule, an MO diagram effectively shows the energetics of the bond between the two atoms, whose AO unbonded energies are shown on the sides. The unbonded energy levels are higher than those of the bound molecule, which is the energetically-favored configuration. Nov 27, 2021 · H2S Molecular Orbital (MO) Diagram.The molecular orbital diagram of H2S can be explained in the following way. This is the MO diagram of H2S. The left-hand side will contain the atomic orbital s of sulfur i.e 3s2 3px2 3py1 3pz1. And on the right-hand side, there will be atomic orbital s of hydrogen. 8 valence electrons are filled in the MO orbital s.
Orbital diagram nitrogen. Draw the resulting d orbital diagram s, labeling energies in terms of es and ep. Answer: Trigonal bipyramidal, ligands are at positions 1, 6, 2, 11, and 12. Trigonal bipyramidal mo diagram. ML4 square planar complexes MO diagram s-only bonding Sample - bonding eg 11. ... As a promising hydrogen storage material, sodium borohydride (NaBH4) exhibits superior stability in alkaline solutions and delivers 10.8 wt.% theoretical hydrogen storage capacity. Nevertheless, its hydrolysis reaction at room temperature must be activated and accelerated by adding an effective catalyst. In this study, we synthesize Co nanoparticles supported on bagasse-derived porous carbon ... Orbital Filling Diagrams. An orbital filling diagram is the more visual way to represent the arrangement of all the electrons in a particular atom. In an orbital filling diagram, the individual orbitals are shown as circles (or squares) and orbitals within a sublevel are drawn next to each other horizontally. The periodic table, also known as the periodic table of (the) chemical elements, is a tabular display of the chemical elements.It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry.It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit a periodic dependence on their atomic numbers.
The orbital diagram of nitryl fluoride, representing only the sigma bonds, is shown below. Therefore, the hybridization of the nitrogen atom in Nitryl Fluoride is sp2 hybridization with trigonal planar geometry . The period of nitrogen is 2 and nitrogen is a p-block element. The electron configuration of nitrogen(N) and the orbital diagram is the main topic of this article. Also, period and group determination, valency and valence electrons of nitrogen, various reactions and compound formation, bond formation of nitrogen have been discussed. The molecular orbital diagram representing this order of energy levels is shown in fig. Fig. No. 5 Order of Energy Levels for Boron, Carbon, Nitrogen etc. This kind of energy reversal is due to mixing of 2s and 2p orbitals where the energy difference is very close, that is, for B, C, and N atoms. According to the symmetry interactions, the two. The optained peptides were analyzed using mass spectrometer with the orbital trap (Orbitrap Elite; Thermo Scientific, Germany). Panoramic spectra were recorded in the m / z range of 300 to 2000 with a resolution of 240,000. An HCD camera was used to perform ion fragmentation. A resolution of 60,000 was adopted to record the fragmentation spectra.
27) An orbital is a probability map showing exactly where an electron can be found in an atom. 28) The higher the principal quantum number, the lower the orbital energy. 29) The possible values for the principal quantum numbers are: . 30) The subshells of the orbital are represented by the possible letters: s, p, d, or f. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of ... Blank diagram of the respiratory system. Jun 4, 2015 - In this assignment, students color the various parts of the respiratory system and the n answer some follow up questions to describe the... The lungs' main function is to help oxygen enter the red cells in the blood. ... The diagram below shows in which part of the respiratory tract some of the...Jan 28, 2019 — This chart of the ... Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the boron group it has three valence electrons for forming covalent bonds, resulting in many compounds such as boric acid, the mineral borax, and the ultra-hard crystal boron carbide.
Then, the ITO-coated glass was dried with nitrogen-flow, and irradiated with oxygen plasma for 20 min. Next, the ZnO precursor solution prepared according to the reported literature was spin-coated on top of the ITO-coated glass with spin-coating rate of 4500 rpm for 30 s. And the above film was annealed at 220 °C for 40 min in air.

1 Create The Atomic Orbital Of Diagram For Nitrogen 2 Construct The Orbital Diagram For Ni Homeworklib
The location of the unpaired electron in a nitrogen sp 2 orbital, and a consequently it forms a dimer through a N-N bond (Figure \(\PageIndex{1}\)e). Furthermore, it is easy to oxidize nitrogen dioxide to the nitronium ion (NO 2 + ), (8.4.11).
Feb 15, 2021 · What is the Orbital Diagram For Nitrogen? When we talk about the orbital diagram, we first need to understand what exactly it means. Therefore, during exams, the student can expect questions related to this topic so it is important that the students must go through it.
The bohr diagram is the diagram of the electrons on the orbital layers of the nucleus of an atom. for potassium, you would put 2 electrons on the first layer, 8 on the second layer, and 9 on the ...Feb 15, 2021 · Ground State Electron Configuration For Nitrogen. When we talk about the electronic configuration, then the ground state Nitrogen Electron Configuration is written as 1s 2 2s 2 2p 3 ...

Give Orbital Diagram Of The Following A Magnesium Chloride B Nitrogen C Methane D Hydrogen Chloride Flash Education
Nowadays, Mn-doping is considered as a promising dissolution for the heavy usage of toxic lead in CsPbX3 perovskite material. Interestingly, Mn-doping also introduces an additional photoluminescence band, which is favorable to enrich the emission gamut of this cesium lead halide. Here, a solution spraying strategy was employed for the direct preparation of CsPbxMn1−x(Br,Cl)3 film through ...

Give Orbital Diagram Of The Following A Magnesium Chloride B Nitrogen C Methane D Hydrogen Chloride Flash Education
Here, one sp orbital of C fuses with 1s orbital of H. And the other sp orbital of C fuses with one of the p orbitals of Nitrogen. The px orbitals of both C and N form sigma bonds while the Py and Pz orbitals form perpendicular Pi bonds. Polarity of HCN. Now let us look at whether the compound is polar or nonpolar in nature.
Fluorine electron configuration is 1s 2 2s 2 2p 5.The symbol for fluorine is F. The period of fluorine is 2 and it is a p-block element. The electron configuration of fluorine(F) and the orbital diagram is the main topic of this article.
Draw The Orbital Diagrams Representing The Atomic Structures Of The Following A Hydrogen B Helium C Lithium D Carbon E Nitrogen Chemistry Topperlearning Com Hmdp8scgg
In writing the electron configuration for Copper the first two electrons will go in the 1s orbital. The next six electrons will go in the 2p orbital. The next six electrons. So for Ru 3 you would first remove 2 electrons from the s orbital and one for the d orbital leading to a configuration of Ar 3d5. To form K 1 we remove electron.
WGS - Wideband Global Satcom WGS Satellites under construction at Boeing - Image: Boeing Space Systems. WGS - The Wideband Global Satcom System is a U.S. satellite constellation providing communications for military operations around the globe, supporting soldiers, ships, and aircraft including unmanned drones used for surveillance and intelligence gathering operations around the globe.
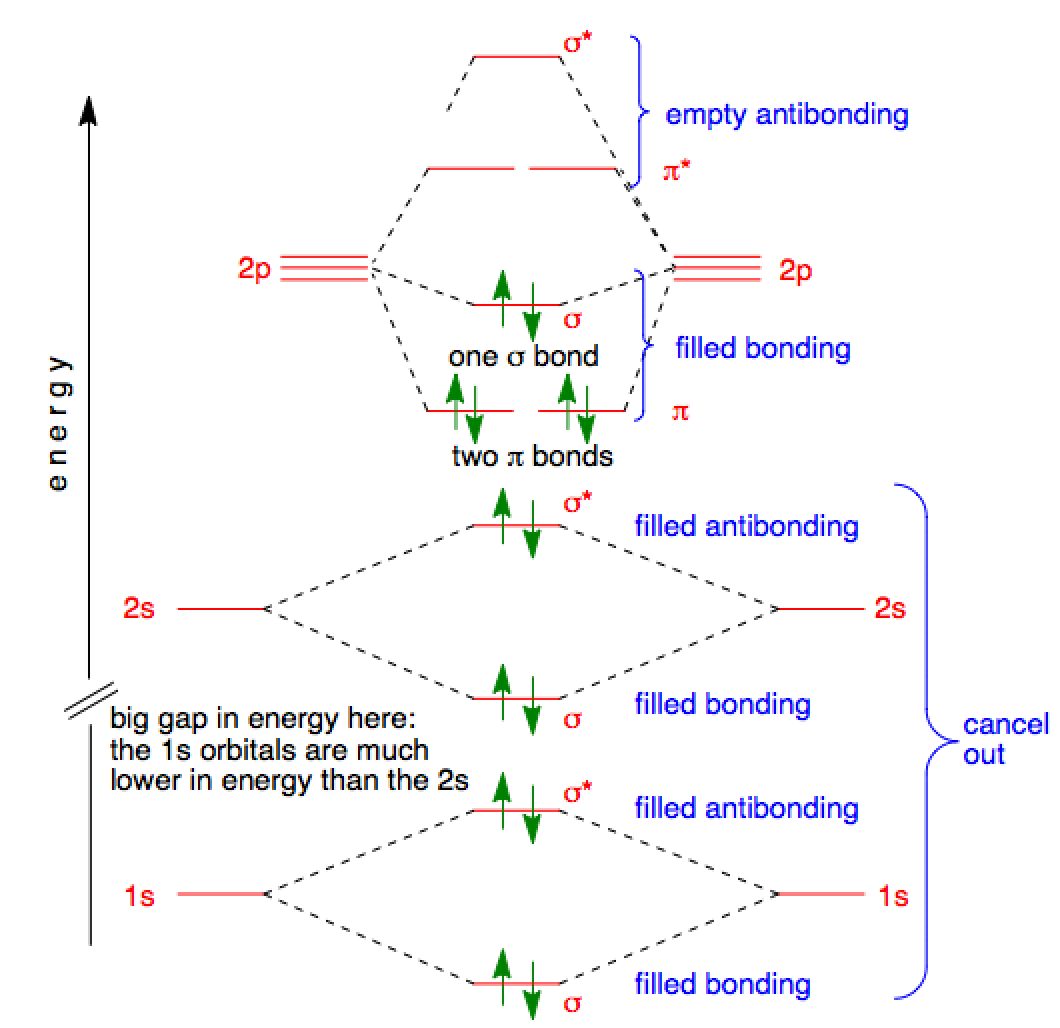
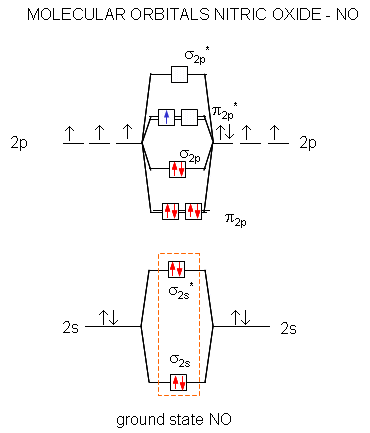
Now the next topic to cover is the molecular orbital diagram of nitrous oxide. N2O Molecular Orbital Diagram. Molecular orbital diagrams say about the mixing of orbitals in a compound. Using a MO diagram, the bond order of a compound can be determined which gives us an idea about bond length, bond stability as well.

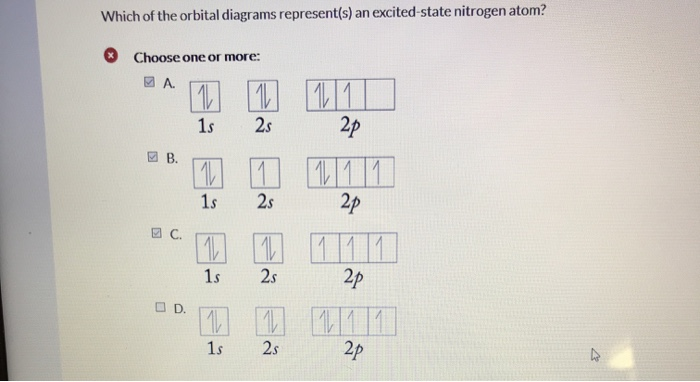
Which Of The Orbital Diagrams Represent S An Excited State Nitrogen Atom Choose One Or More 1 2p Homeworklib
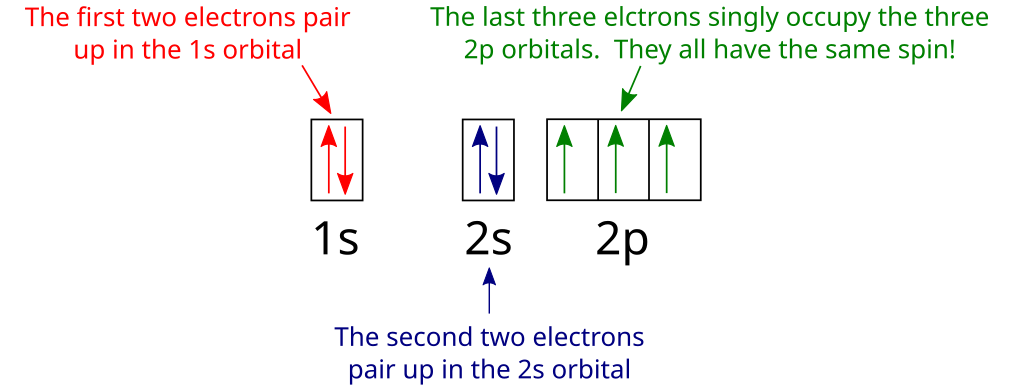
A nitrogen atom has seven electrons. In the ground state, they are arranged in the electron configuration 1s 2 2s 2 2p 1 x 2p 1 y 2p 1 z.It therefore has five valence electrons in the 2s and 2p orbitals, three of which (the p-electrons) are unpaired. It has one of the highest electronegativities among the elements (3.04 on the Pauling scale), exceeded only by chlorine (3.16), oxygen (3.44 ...
Feb 23, 2016 · The orbital filling diagram for helium. The electron configuration for helium is 1s². This means that we have two electrons in the 1s orbital, which looks like this: This diagram is exactly the same as the one for hydrogen, except that there’s a second arrow added to the 1s orbital.
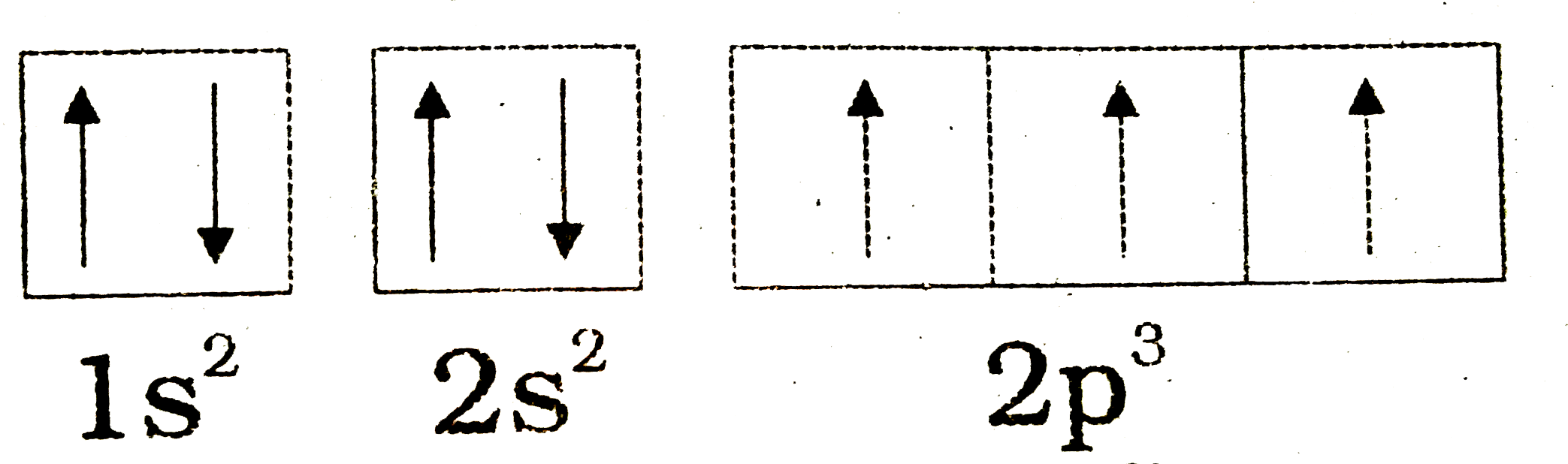
Following Orbital Diagram Shows The Electronic Configuration Of Nitrogen Atom Which Rule Does Not Support This Img Src Https D10lpgp6xz60nq Cloudfront Net Physics Images Brs Qb Phy Sci X C06 E01 017 Q01 Png Width 80
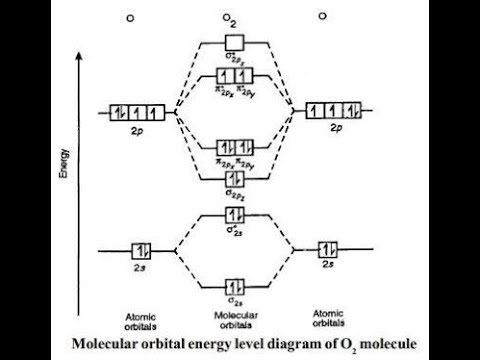
A molecular orbital diagram or mo diagram is a qualitative descriptive tool explaining mo diagrams can explain why some molecules exist and others do not. Draw the molecular orbital diagram for the molecular ion n 2. Individual atomic orbitals ao are arranged on the far left and far right of the diagram. Carbon monoxide molecular orbital diagram.
1.0 Pa (2015) Composition by volume. Nitrogen, methane, carbon monoxide. Pluto compared in size to the Earth and Moon. Pluto ( minor-planet designation: 134340 Pluto) is a dwarf planet in the Kuiper belt, a ring of bodies beyond the orbit of Neptune. It was the first and the largest Kuiper belt object to be discovered.
NF3 Lewis Structure, Molecular Geometry, Hybridization, Polarity, and MO Diagram. Nitrogen trifluoride or NF3 is a nitrogen halide compound that is slightly water-soluble. Its noticeable characteristics include being colorless and carrying a musty or moldy odor. NF3 has a molar mass of around 71.002 g/mol and a density of 3.003 kg/m3.
Mar 04, 2021 · The lithium 1s orbital is the lowest-energy orbital on the diagram. Because this orbital is so small and retains its electrons so tightly, it does not contribute to bonding; we need consider only the 2 s orbital of lithium which combines with the 1 s orbital of hydrogen to form the usual pair of sigma bonding and antibonding orbitals.
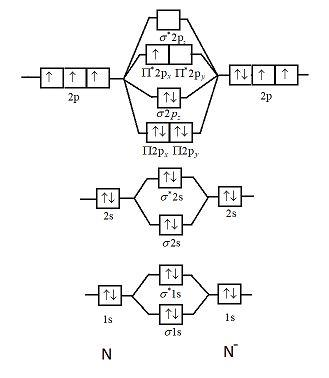
Procedure to draw the molecular orbital diagram of CN. 1. Find the valence electron of each atom in the CN molecule. Clearly, carbon has 4 valence electrons and nitrogen has 5. 2. Find if the molecule homo-nuclear diatomic molecular orbital or hetero-nuclear diatomic molecular orbital. Clearly, CN is hetero orbital. 3.
Volvo v70 evap system diagram. Here is the Volvo Vida's information. However I threw out the other list of partnumbers for this diagram for part numbers. However if these are dealer parts your gonna pay like $30 a hose. I'm unsure of the sizes, they are prebent hoses and getting the angles right using a regular radiator hose won't work well.
Orbital energy diagram s are provided to guide you learn about the atomic orbital.A chemical equation shows the chemical formulas of substances that are reacting and the substances that are produced. Depict the electron configuration for magnesium ing an orbital box diagram and noble gas notation. So this question is asking us to construct an orbital diagram to show the electron configuration ...
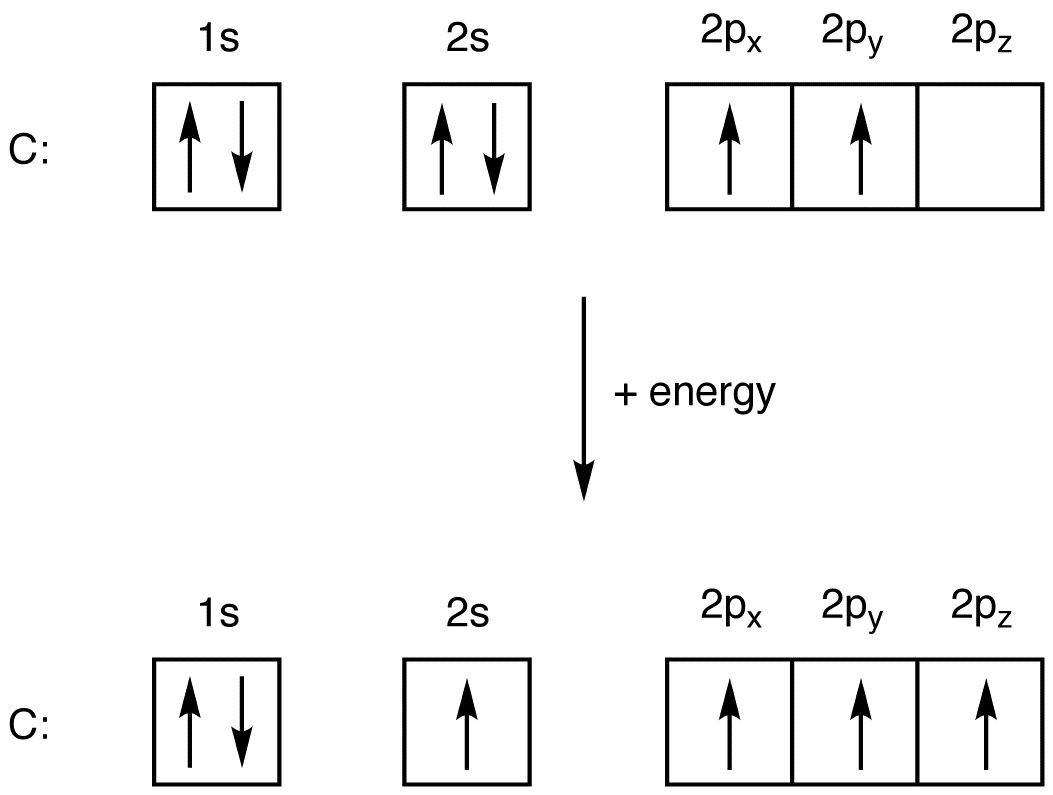
The atomic number of Carbon is 6 so 2 electrons are filled in s orbital and the rest 4 are in the outer orbital that is why the valence number of electrons in carbon is 4. For Nitrogen, its atomic number is 7, so after 2 electrons occupy s orbital, the rest 5 are in the outer orbital so the valence number of electrons is 5.
Nov 27, 2021 · H2S Molecular Orbital (MO) Diagram.The molecular orbital diagram of H2S can be explained in the following way. This is the MO diagram of H2S. The left-hand side will contain the atomic orbital s of sulfur i.e 3s2 3px2 3py1 3pz1. And on the right-hand side, there will be atomic orbital s of hydrogen. 8 valence electrons are filled in the MO orbital s.
Molecular orbital diagram for hydrogen: For a diatomic molecule, an MO diagram effectively shows the energetics of the bond between the two atoms, whose AO unbonded energies are shown on the sides. The unbonded energy levels are higher than those of the bound molecule, which is the energetically-favored configuration.
An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. A box, line, or circle, is drawn to represent each orbital in the electron configuration (using the Aufau Principle to order the orbitals and hence the boxes, lines or circles, as shown below)

Solved Draw The Molecular Orbital Diagram For The Cyanide Ion Cn1 Versus The Nitrogen Atom N2 A Molecular Orbital Diagrams B Bond Order For Each Molecule C Molecular Orbital Configuration For The
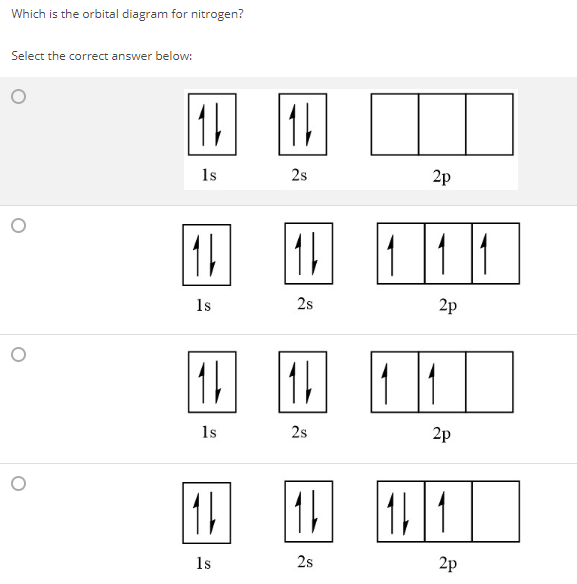
Solved Write The Full Electron Configuration Fill In The Orbital Diagrams And Determine The Magnetic Property For The Following Elements Nitrog Course Hero

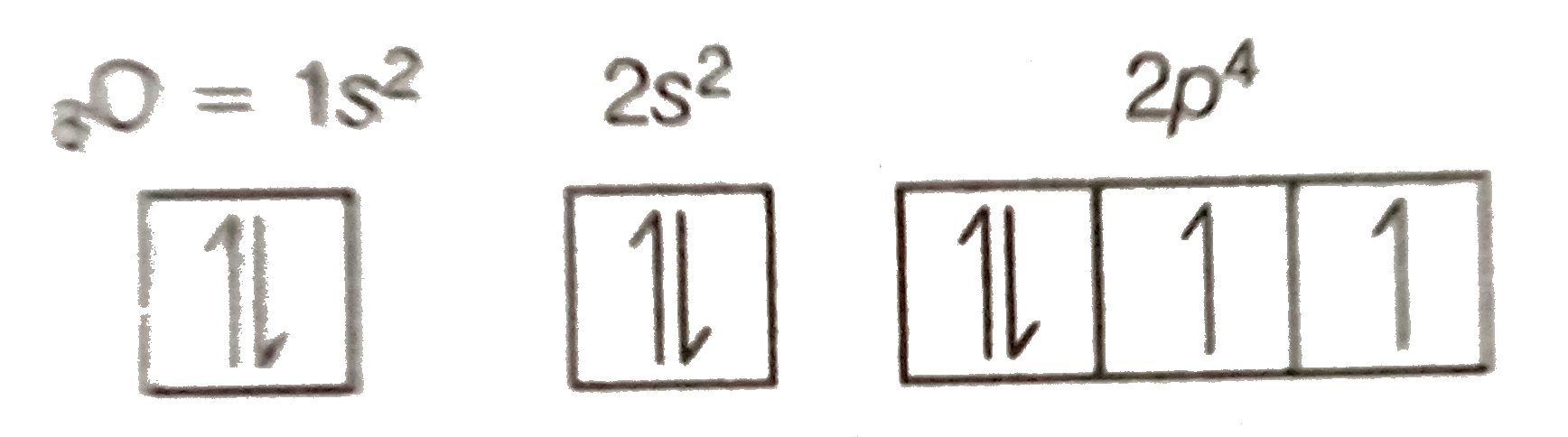







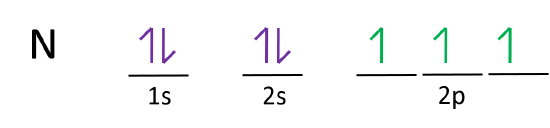






Comments
Post a Comment