42 lewis dot diagram for sodium chloride
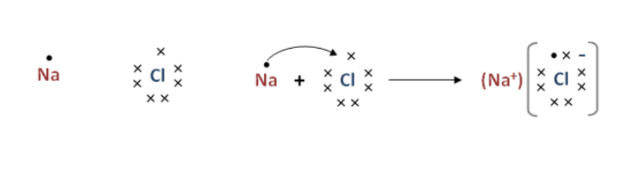
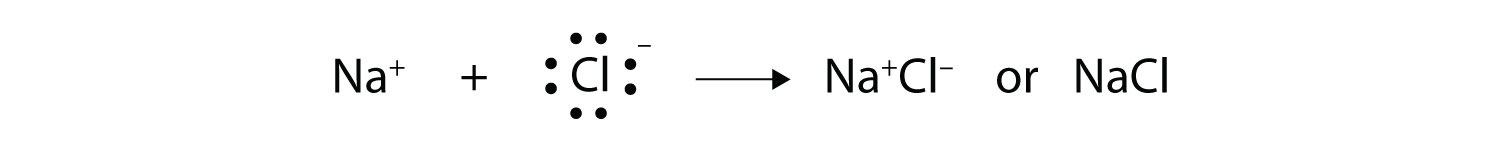
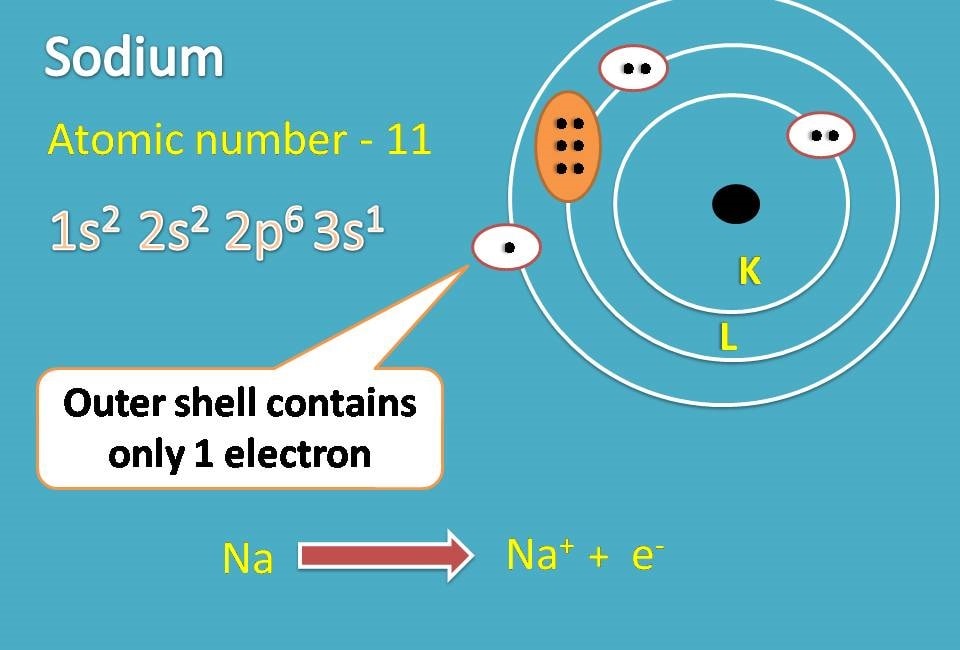
The Lewis dot structure for water shows the electron from hydrogen and an electron from oxygen being shared in a covalent bond. ... Ionic Bonding of Sodium Chloride. When sodium loses its only valence electron to become an ion, the Lewis structure shows it with no dots (electrons). Sodium, a metal in Group 1, loses one electron to become a +1 ionChlorine, a non-metal in Group 17, gains one electron to become a -1 ion.Together, they comb...
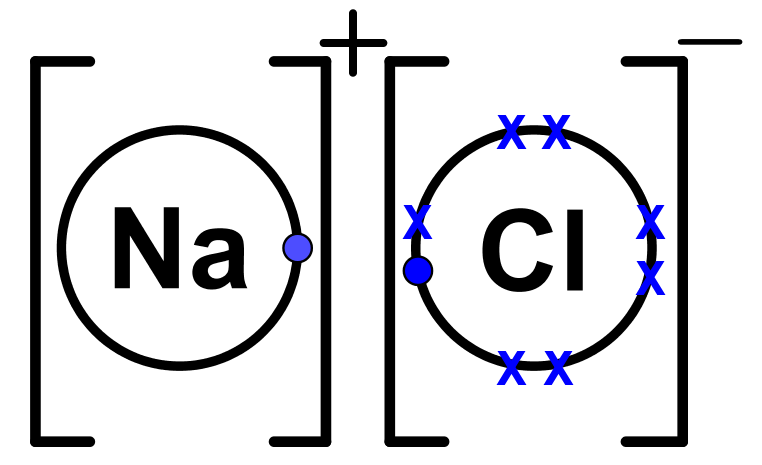
The lewis dot structure of nacl consists of a chloride ion surrounded by eight electron dots four pairs and a sodium ion bonded to that chlorine ion. Although these electron arrangement diagrams show how the ionic bond is formed and the electronic structure and electrical charge on the ions they do not give any idea on the relative size of the ...

Lewis dot diagram for sodium chloride
Chemical BONDING IONIC Lewis Dot Diagrams Sodium Chloride This is the finished Lewis Dot Structure [Na]+1 [ Cl ]-1 How did we get here? Practice Dot diagrams & formulas Lithium fluoride Magnesium oxide Calcium chloride Potassium hydride Drawing molecules using Lewis Dot Structures Remember: atoms are sharing e- to complete their outer shell! A step-by-step explanation of how to draw the NaCl Lewis Dot Structure (Sodium chloride).For NaCl we have an ionic compound and we need to take that into acc... Draw the lewis dot structure for mgi2. Draw the Lewis Dot structure for RbIO2. Lewis Dot Structures of Atoms and Ions. Rubidium iodide crystallizes with the same structure as sodium chloride. Include all hydrogen atoms and nonbonding electrons. A step-by-step explanation of how to draw the LiCl Lewis Dot Structure. Show the formal charges of ...
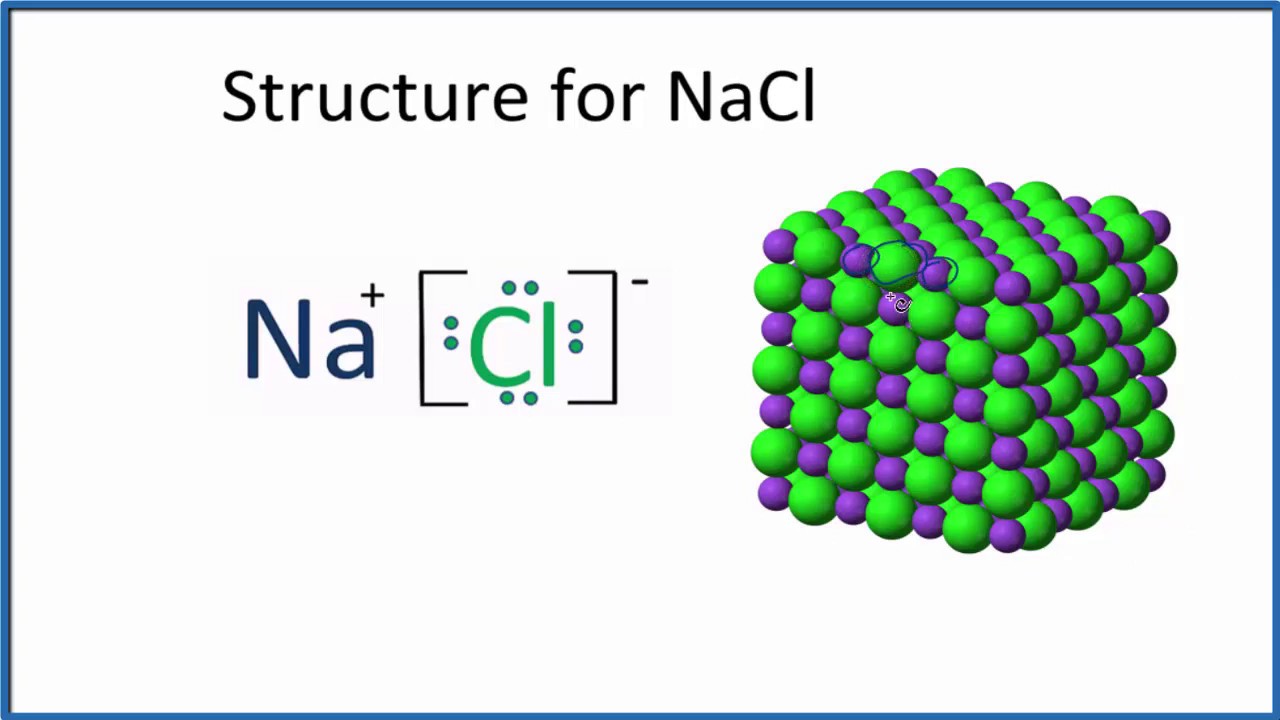
Lewis dot diagram for sodium chloride. A. The Lewis dot structure for francium up with chlorine's unpaired dot. Chemists often depict a bond with a line, so sodium chloride can be written as Na -Cl. Draw the correct Lewis dot structure for CH2O & determine the shape Trigonal Planar. A sodium ion (2,8)+. Diagram of bonding in sodium chloride. A sodium atom gives an electron to a ... Draw electron dot representation for the formation of sodium chloride. Medium. Open in App. Solution. Verified by Toppr. The structure of dot representation for the formation of NaCl is written above. Was this answer helpful? 0. 0. Similar questions. Write Lewis symbols for the following atoms and ions : (i) ... dot diagrams. Lewis dot diagrams will continue to be useful throughout the unit, and the semester. as students are introduced to covalent bonding and continue to recognize periodicity in whether atoms gain, lose or share electrons to form bonds. Ninth grade Lesson Ionic Bonding With Lewis Dot Diagrams How Ionic compounds are formed in the form ... The total valence electron is available for drawing the Sodium chloride (NaCl) lewis structure is 8. NaCl is a face-centered cubic unit cell that has four cations and four anions. In the NaCl lewis dot structure, the sodium atom completes its octet by transferring one electron to the chlorine atom.
The lewis dot structure of nacl consists of a chloride ion surrounded by eight electron dots four pairs and a sodium ion bonded to that chlorine ion. In the case of the sodium cation the filled shell is the outermost of the core electron shells. A step-by-step explanation of how to draw the Cl- Lewis Dot Structure.For the Cl- structure use the periodic table to find the total number of valence electr... A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the ... Na (sodium), a metal, loses one electron to become a +1 ionCl (chlorine), a non-metal, gains on electron to become a -1 ion.Together, they form NaCl, one uni...
The Lewis Diagram for a Sodium ion is shown to the left. Since Sodium is a metal, it has relatively low values for ionization energy and electronegativity. This means that sodium loses an electron to achieve the stable noble gas configuration of 8 valence electrons. We must take away 1 dot from the dot diagram of the sodium atom and put in the resulting charge of +1. 28 Lewis Dot Diagram For Nacl - Wiring Diagram List Draw the electron dot structures of sodium and chlorine Show the ... Lewis Dot Diagram For Nacl - Diagram Resource Gallery Check me out: http://www.chemistnate.com Using Lewis dot structures and the octet rule, we can predict and represent the electronic structure of covalently bonded molecules. For example, when two chlorine atoms, each with 7 valence electrons, come together to form a diatomic chlorine molecule, the Lewis structure shows that there will be a sharing of two electrons between the two ...
The Lewis dot structure of NaCl consists of a chloride ion surrounded by eight electron dots (four pairs) and a sodium ion bonded to that chlorine ion. Typically, ionic Lewis dot structures include the ionic charge, so the Na ion is labeled +1 and Cl is labeled -1. The Lewis dot structure of ionic bonds such as NaCl is formed by looking at both ...
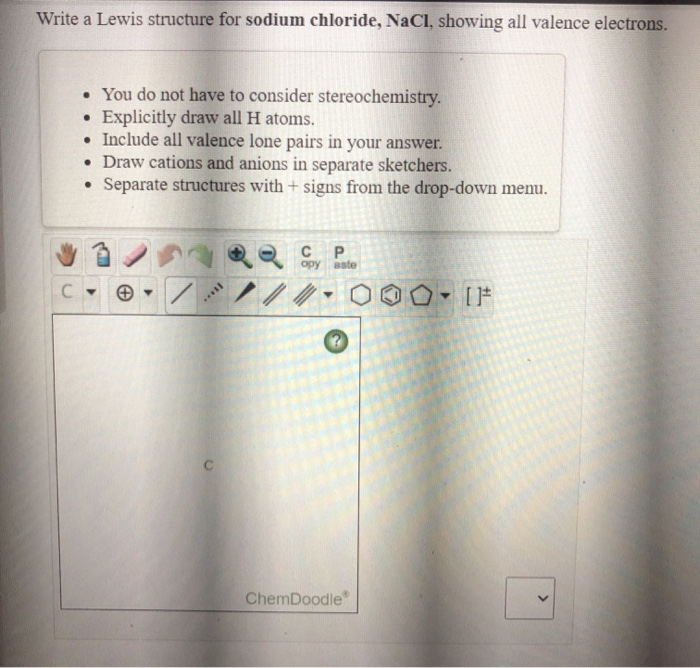
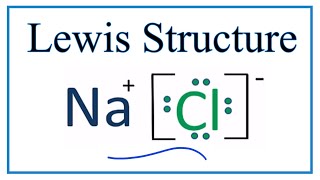
A step-by-step explanation of how to draw the NaCl Lewis Dot Structure.For the NaCl Lewis structure, calculate the total number of valence electrons for the ...
How Ionic compounds are formed in the form of an animation!Enjoy!NOTE: The Lewis Dot Diagram in the end is the one for Sodium Chloride.Don't forget to subscr...
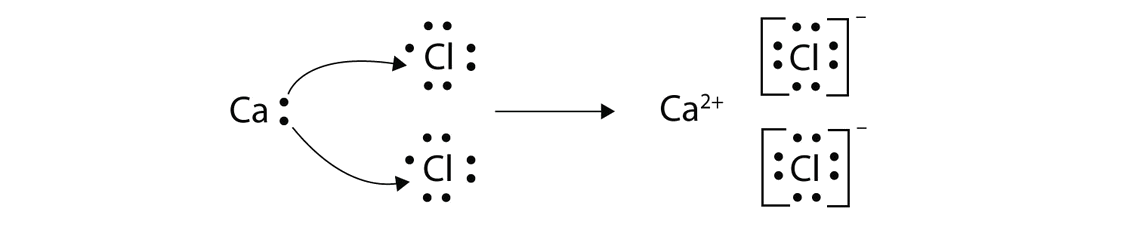
This is the correct dot diagram for sodium, group 1. Q. This is the correct dot diagram for nitrogen, group 15. Q. This is a correct dot diagram for neon, group 18. Q. This could be the dot diagram of. Mg, group 2. Cl, group 17. Magnesium lewis dot diagram. It is MgBr2. Magnesium has an electronic configuration of 2.8. 2.
Lewis dot structure chemistry worksheet answers. You have remained in right site to start getting this info. 8 c2h5oh ethanol 9 n2f4. Be sure you know how to draw correct lewis dot structures and are able to correctly predict the electronic arrangement and molecular geometry before going on to the lab assignment. ... Sodium chloride magnesium ...
Fill in the lewis dot symbols for. Sodium reacts explosively with chlorine gas to form sodium chloride. Lewis dot structures allow us to understand two types of bonding ionic and covalent. Some of the worksheets for this concept are chemical bonds lewis dot structures work practice problems h s so ch br hcn bonding basics bonding basics ...
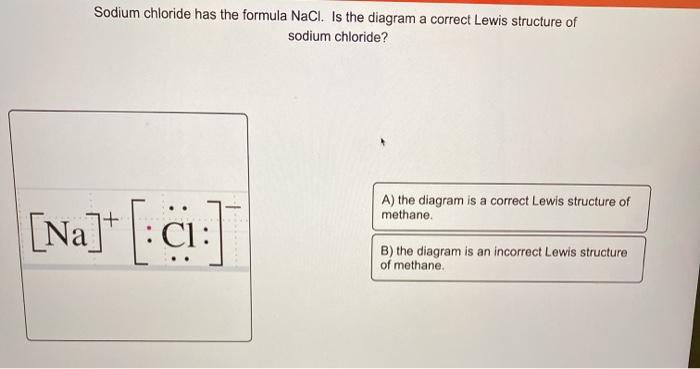
What is the correct Lewis dot structure for Sodium chloride? Lewis structure of HCN looks like... Lewis Dot Structure. diagram of a molecule using dots to represent valence electrons. octet rule. atoms react by gaining or losing electrons so as to acquire the stable electron structure of a noble gas, usually eight valence electrons.

Explain The Formation Of Sodium Chloride With Help Of Electron Dot Structure At Numbers Na 11 Brainly In
What is the Lewis dot structure for sodium? As you can see Chlorine is now surrounded by 8 electrons in the n=3 shell and Sodium has lost its one valence electron in the n=3 shell. Of course, Sodium, is still surrounded by the 8 electrons of the n=2 shell, but we do not show electrons in the inner closed shells….Lewis Dot Structures.
Since sodium is a metal, it has relatively low values for ionization energy and electronegativity. This means that sodium loses an electron to achieve the stable noble gas configuration of 8 valence electrons. We must take away 1 dot from the dot diagram of the sodium atom and put in the resulting charge of +1.
Draw the lewis dot structure for mgi2. Draw the Lewis Dot structure for RbIO2. Lewis Dot Structures of Atoms and Ions. Rubidium iodide crystallizes with the same structure as sodium chloride. Include all hydrogen atoms and nonbonding electrons. A step-by-step explanation of how to draw the LiCl Lewis Dot Structure. Show the formal charges of ...
A step-by-step explanation of how to draw the NaCl Lewis Dot Structure (Sodium chloride).For NaCl we have an ionic compound and we need to take that into acc...
Chemical BONDING IONIC Lewis Dot Diagrams Sodium Chloride This is the finished Lewis Dot Structure [Na]+1 [ Cl ]-1 How did we get here? Practice Dot diagrams & formulas Lithium fluoride Magnesium oxide Calcium chloride Potassium hydride Drawing molecules using Lewis Dot Structures Remember: atoms are sharing e- to complete their outer shell!
Give The Electron Dot Structures Of I Nacl Ii Mgcl2 Iii Cao Sarthaks Econnect Largest Online Education Community

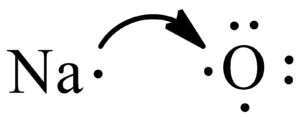
Write The Electron Dot Structure For Sodium And Chlorine Atoms How Do These Form A Chemical Bond Brainly In

Ionic Bonding In Sodium Chloride When Only The Outer Electron Shells Are Drawn Ionic Bonding Vimeo Logo Tech Company Logos











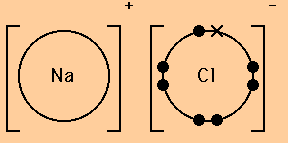








Comments
Post a Comment