38 co2 electron dot diagram
Structure of SO 3: It is a D3h symmetry trigonal planar molecule in its gaseous state, as predicted by VSEPR theory. As a result, it is said to be a member of the D3h point group. According to the electron-counting formalism, the sulphur atom has an oxidation state of +6 and a formal charge of 0. Interplay between local structure and transport properties in. Posted on 30.11.2021 by fekag. Getting to and away from the egg, an interplay between ...
Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes. SAVE IMAGE. 11 Na Sodium Electron Shell Structure Schoolmykids Matter Worksheets Element Chemistry Electron Configuration.

Co2 electron dot diagram
Dot-and-cross diagrams including molecular ions • The molecular ions have covalent bonds in but have gained or lost electrons from the elements within them. • That means they bind ionically to other oppositely charged ions: • Calcium hydroxide (Qu10). 2 Clue for Q9: Cyanide is a molecular ion: - CΞN H O - Ca 2+ In this article, we will discuss Phosphorous trifluoride (PF3) lewis dot structure, molecular geometry, electron geometry, hybridization, polar or nonpolar, its bond angle, etc. " Phosphorus trifluoride is similar to carbon monoxide in that it is a gas which strongly binds to iron in hemoglobin, preventing the blood from absorbing oxygen." 6 Jun 2020 — There are a total of 40 valence ... Fred Lambert. - Nov. 19th 2021 12:26 pm PT. @FredericLambert. Electrek Podcast. This week on the Electrek Podcast, we discuss the most popular news in the world of sustainable transport and energy ...
Co2 electron dot diagram. One return trip out there from the UK costs 1.96 tons of what the scientists call carbon dioxide equivalent - the bad stuff. Any worries about whether or not Sir Geoffrey should be doing some private work while also trying to serve his constituents is suddenly dwarfed by the damage to the planet, just like a bigger version of my drive to ... CO2 adsorption properties of the CuTATB-n were measured at 273 and 298 K, which indicated the contribution of catenation in CuTATB-n for CO2 capture. The adsorption isotherms for CO2 and N2 show high adsorption capacity for CO2 (189 mg g-1 and 156 mg g-1, resp. for CuTATB-60 and -30 at 298 K) and excellent selectivity over N2 (>20:1). Bonding Energy Lewis Structures and VSEPR Worksheet 1. 2 For each of the following compounds a Lewis structure determine the bond angles and molecular shapes for all atoms. Lewis Structure And Vsepr Models. This information given a dot structure and lewis vsepr worksheet with. H2O 8 4 Tetrahedral Bent CO2 G-NH3 5-3 BF3. 2. Man only emits 6% of total annual CO2 emissions (Nature emits 94%). Add the numbers on the NASA diagram at NASA's earth observatory Cabon Cycle diagram 3. CO2 only causes 26-32% of the greenhouse effect. (H2O is 60-75%)
Zinc oxide is an inorganic compound with the formula Zn O.ZnO is a white powder that is insoluble in water. It is used as an additive in numerous materials and products including cosmetics, food supplements, rubbers, plastics, ceramics, glass, cement, lubricants, paints, ointments, adhesives, sealants, pigments, foods, batteries, ferrites, fire retardants, and first-aid tapes. The major greenhouse gases in our atmosphere, water vapour 1st and carbon dioxide 2nd, are both many doublings beyond saturation (about 10 doubling for CO2 - at 400 ppm, CO2 absorption for the atmospheric column at line centre is around 3000 absorbance). One additional doubling should give about 1/10 of the total impact of CO2. nitrogen dioxide专题🌟整理关于💖相关图片资讯希望大家喜欢。 NO2 nitrogen dioxide molecule Royalty Free Vector Image A covalent compound is a chemical compound that is made when two nonmetals or two p-block elements together share their valence electron with each other to complete the octet and attains stability. is no2 ionic or covalent. The sharing of valence electrons between two atoms makes a bond which is called a covalent bond.
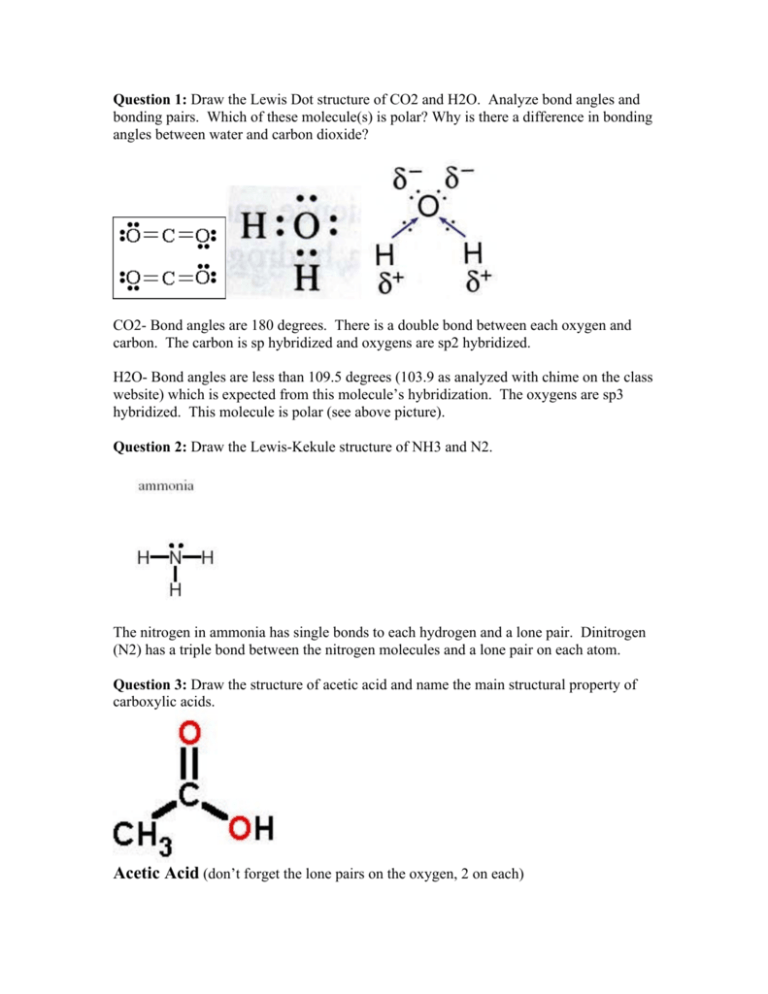
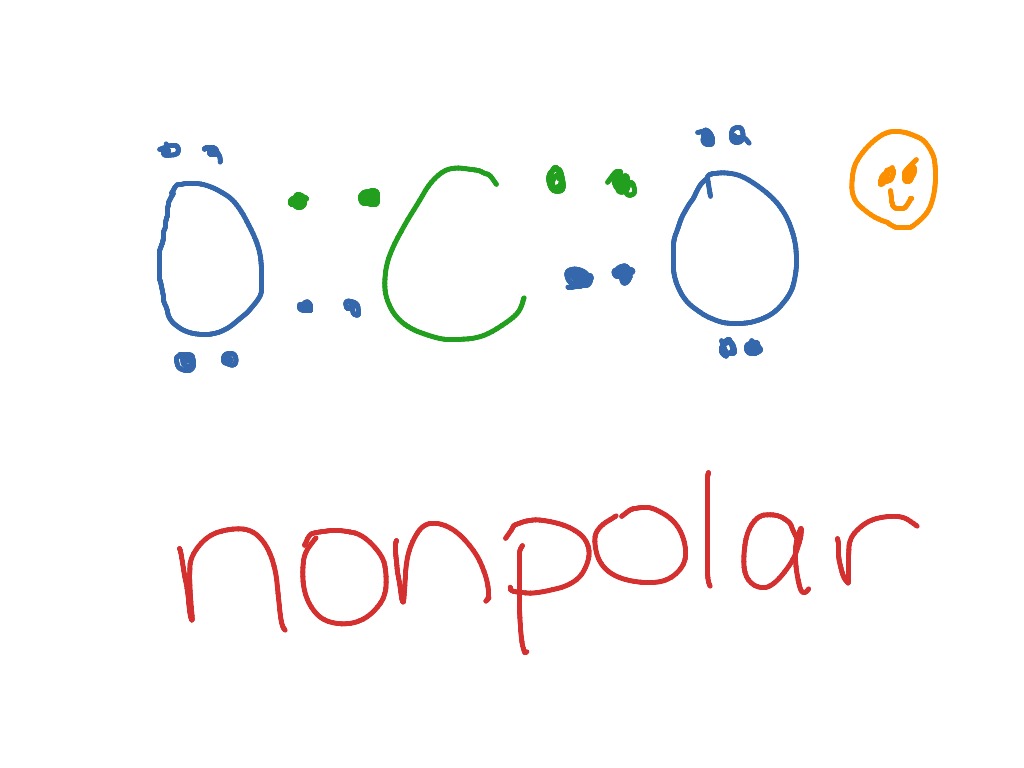
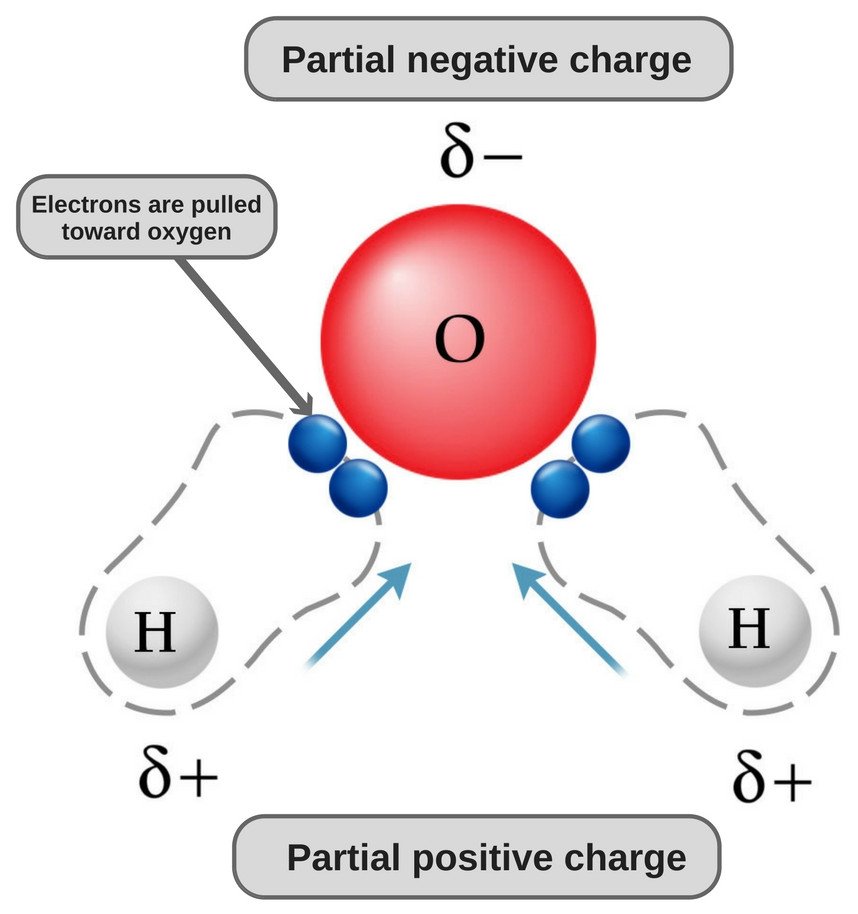
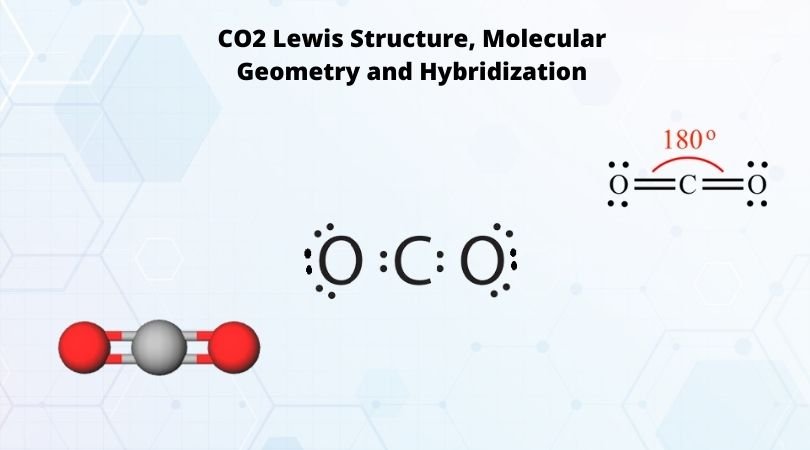
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula ‹The template Silicon is being considered for deletion.› Si O 2, most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand.Silica is one of the most complex and most abundant families of materials, existing as a compound ... [ASAP] Superhydrophobic Silica Aerogels and Their Layer-by-Layer Structure for Thermal Management in Harsh Cold and Hot Environments ACS Nano DOI: 10.1021/acsnano.1c07184 Electron Dot Diagrams Determining Important Bonding Information Chemical Bonds - Analogies How Will They Bond? Examining Molecules and Compounds Understanding Chemical Formulas - Subscripts, Parentheses, Coefficients Totaling Atoms Using PSEC (PPT.) Analyzing Chemical Formulas Analyzing Chemical Equations Balancing Chemical Equations Lewis dot poly-atomic molecule, C O X 2 \ce{CO_{2}} C O X 2 . The octet rule for oxygen and the most energetically stable configuration for CO2 is achieved by forming double bonds with each carbon. EXAMPLE. You Try It. Draw the Lewis structure for water H X 2 O \ce{H_{2}O} H X 2 O. Write the letters of the elements you will draw electrons around.

Co2 Lewis Structure Lewis Dot Structure For Co2 Carbon Dioxide Lewis Structure Practices Worksheets Chemistry Worksheets Chemistry Lessons
Hackaday Podcast Ep 146: Dueling Trackballs, Next Level BEAM Robot, Take Control Of Your Bench, And Green Programming Postpone your holiday shopping and spend some quality time with editors Mike ...
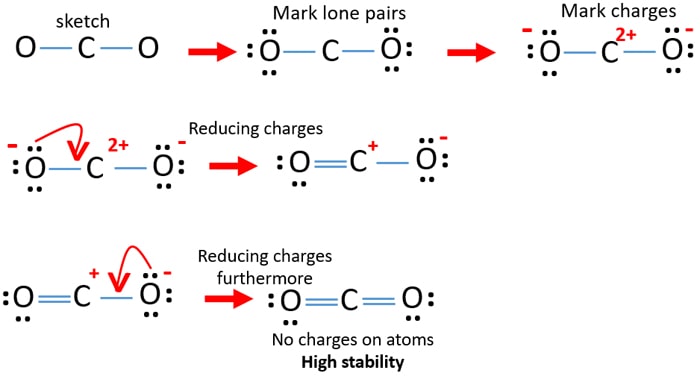
CO2 Lewis Structure. The lewis structure of CO2 can be with some simple steps, but before that, it is important to understand lewis structure properly. So lewis structure generally gives us an idea about the nature of bonding and octet fulfillment of the atoms. According to the octet rule, an atom attains stability by fulfilling its octet.
Lewis structure is also known as electron dot structure or Lewis dot structure because the valence electrons are represented as dots in the Lewis structure of the molecule. It is the two-dimensional structure in which every atom in the molecule tends to complete its octet either by sharing or gaining or losing electrons.
2.11 Quiz: Electron Orbitals. What is the maximum number of electrons that can occupy a box in an orbital filling diagram at any energy level? What is the maximum number electrons that can occupy any d orbital? Use an aufbau diagram. The next two electrons go in to each of p orbital s, with both electrons hav in g the same sp in.
The pf3 lewis structure is one example of a tricapped trigonal bipyramid where there are two sets of parallel planes which intersect at angles not quite 90 degrees so as to form 3-sided pyramids. Pf3 lewis structures are one of the many ways to depict the bonding in a mo lecule. A) trigonal bipyramidal B) ...
Group 16 elements: The Oxygen Family. The modern periodic table's group 16 elements are composed of five elements: oxygen, sulphur, selenium, tellurium, and polonium. Because many elements can be extracted from sulphide or oxide ores, the elements in this group are also known as chalcogens or ore-forming elements. Oxygen is abundant on the ...
Get notifications on updates for this project. Get the SourceForge newsletter. Get newsletters and notices that include site news, special offers and exclusive discounts about IT products & services.
Please feel free to start a scientific chemistry discussion here! Discuss chemistry homework problems with experts! Ask for help with chemical questions and help others with your chemistry knowledge! Moderators: expert, ChenBeier, Xen. 4945 Topics. 11503 Posts. Last post Re: esterification reactions. by raghavan. Sat Nov 27, 2021 10:20 pm.
Fred Lambert. - Nov. 19th 2021 12:26 pm PT. @FredericLambert. Electrek Podcast. This week on the Electrek Podcast, we discuss the most popular news in the world of sustainable transport and energy ...
In this article, we will discuss Phosphorous trifluoride (PF3) lewis dot structure, molecular geometry, electron geometry, hybridization, polar or nonpolar, its bond angle, etc. " Phosphorus trifluoride is similar to carbon monoxide in that it is a gas which strongly binds to iron in hemoglobin, preventing the blood from absorbing oxygen." 6 Jun 2020 — There are a total of 40 valence ...
Dot-and-cross diagrams including molecular ions • The molecular ions have covalent bonds in but have gained or lost electrons from the elements within them. • That means they bind ionically to other oppositely charged ions: • Calcium hydroxide (Qu10). 2 Clue for Q9: Cyanide is a molecular ion: - CΞN H O - Ca 2+

What Would Be The Electron Dot Structure Of Carbon Dioxide Which Has The Formula Co2 Carbon And Its Compounds Science Class 10

Draw The Lewis Dot Structure For Co2 Determine The Electron Geometry And Molecular Shape Of This Molecule Is This Molecule Polar Or Nonpolar Study Com

Draw The Electron Dot Structures Of The Following Compounds And State The Type Of Bonding In Each Case Co2 Mgo H2o Hcl Mgcl2 Flash Education
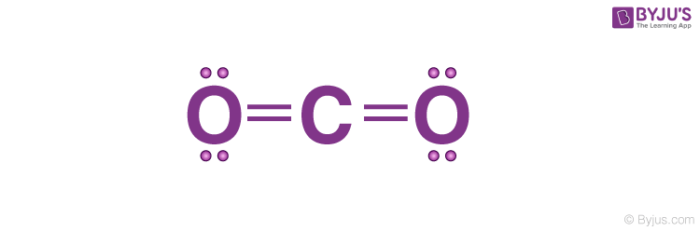
What Would Be The Electron Dot Structure Of Carbon Dioxide Which Has Formula Co Sub 2 Sub Chemistry Q A


/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)


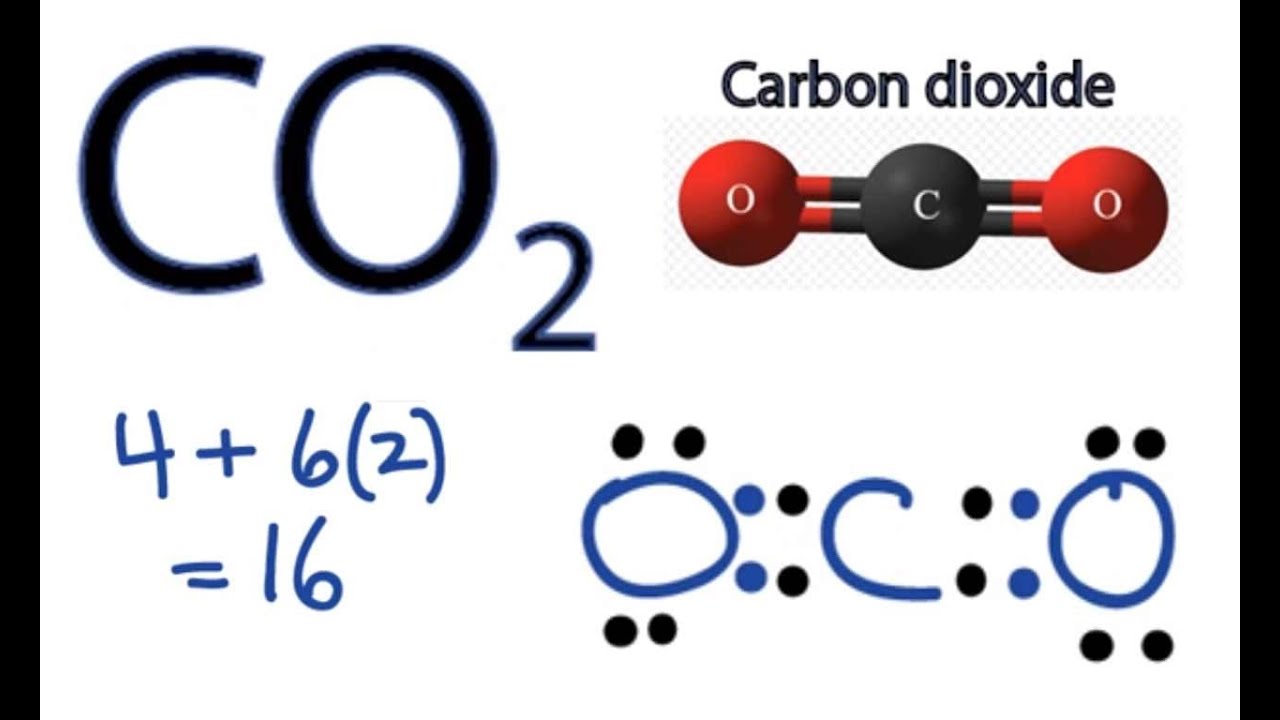


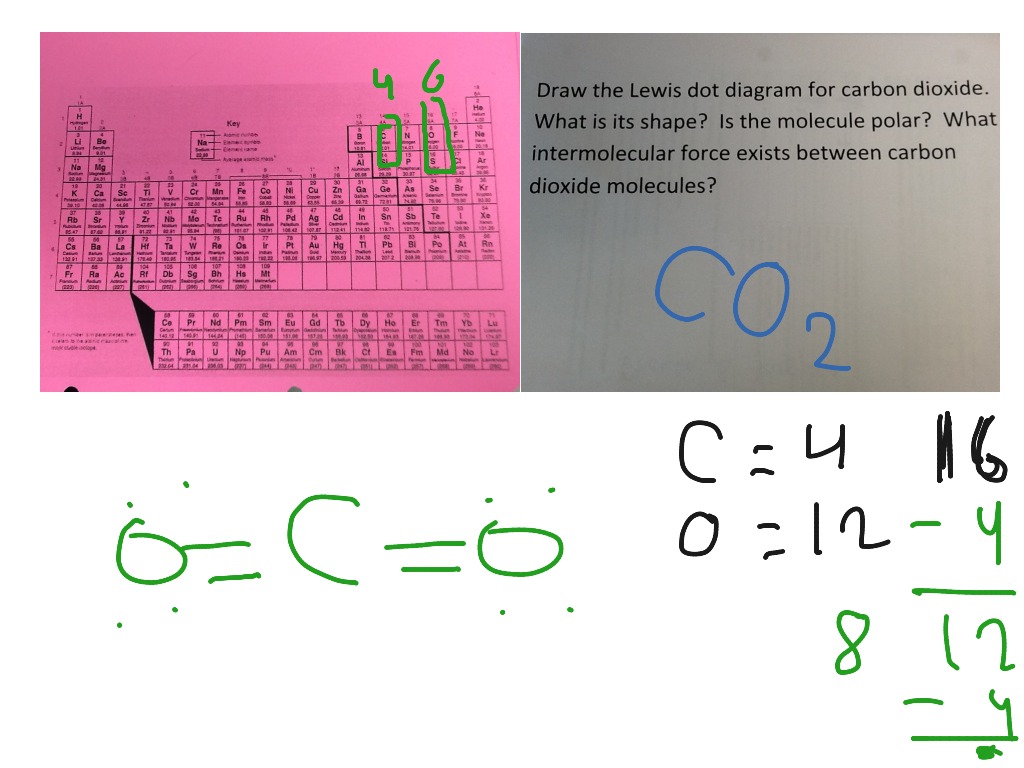












Comments
Post a Comment